





DOI: https://doi.org/10.37349/ebmx.2023.00001

Translating biomaterials research into clinical products is a multidisciplinary yet rewarding journey. It should primarily aim to improve patient treatment and clinical outcomes by addressing unmet clinical needs. Four examples of commercial development of biomaterial implants illustrate the diversity of paths, starting from academic research work (AlchiMedics, TISSIUM, Cousin Surgery) or corporate initiatives to develop new products (Medtronic-Sofradim Production). They have been selected from the Translational Research session of the 2022 Conference of the European Society for Biomaterials (ESB) in Bordeaux (France). Commitment, agility, and perseverance were among the key common skills to successfully meet challenges, especially the most unexpected ones. All dimensions of translation projects must be integrated from the start, including the regulatory strategy.
Translating biomaterials research into clinical products is a multidisciplinary yet rewarding journey. It should primarily aim to improve patient treatment and clinical outcomes by addressing unmet clinical needs. Four examples of commercial development of biomaterial implants illustrate the diversity of paths, starting from academic research work (AlchiMedics, TISSIUM, Cousin Surgery) or corporate initiatives to develop new products (Medtronic-Sofradim Production). They have been selected from the Translational Research session of the 2022 Conference of the European Society for Biomaterials (ESB) in Bordeaux (France). Commitment, agility, and perseverance were among the key common skills to successfully meet challenges, especially the most unexpected ones. All dimensions of translation projects must be integrated from the start, including the regulatory strategy.
DOI: https://doi.org/10.37349/ebmx.2024.00004

Aim:
Small defects after any injury to the periperal nerves results in self-regeneration. However, for larger defects, suturing or grafting are necessary, which may have limitations. Thus, research on nerve guidence conduits is needed without drawbacks. The aim of the study was to develop hydrogel-based conduits containing interpenetrating network (IPN).
Methods:
Methacrylated gelatin (GelMA)-methacrylated hyaluronic acid (HaMA) IPN was filled the poly(2-hydroxyethylmethacrylate) (pHEMA) the outer conduit. Schwann cells (SCs) were used on the pHEMA and the distal end of the tube was injected with netrin-1 to support model SH-SY5Y cells.
Results:
1H-nuclear magnetic resonance (1H-NMR) showed that methacrylation degrees were 94% ± 2% for GelMA and 60% ± 7% for HaMA. The fraction of HaMA increased the degradation rate; pure HaMA degraded in 3 weeks, while pure GelMA in more than 5 weeks. An increase in the fraction of 2-hydroxyethylmethacrylate (HEMA) from 20% to 56% decreased the porosity and the pore size, significantly. SH-SY5Y cells migrated along the conduit in the presence of netrin-1. NeuN expression was increased in 2 weeks indicating neuronal activity.
Conclusions:
SH-SY5Y cells produced neurites in the IPN. pHEMA conduit including GelMA-HaMA IPN is a good candidate for peripheral nerve regeneration applications. As future studies, the conduit will be tested in vivo for nerve regeneration.
Aim:
Small defects after any injury to the periperal nerves results in self-regeneration. However, for larger defects, suturing or grafting are necessary, which may have limitations. Thus, research on nerve guidence conduits is needed without drawbacks. The aim of the study was to develop hydrogel-based conduits containing interpenetrating network (IPN).
Methods:
Methacrylated gelatin (GelMA)-methacrylated hyaluronic acid (HaMA) IPN was filled the poly(2-hydroxyethylmethacrylate) (pHEMA) the outer conduit. Schwann cells (SCs) were used on the pHEMA and the distal end of the tube was injected with netrin-1 to support model SH-SY5Y cells.
Results:
1H-nuclear magnetic resonance (1H-NMR) showed that methacrylation degrees were 94% ± 2% for GelMA and 60% ± 7% for HaMA. The fraction of HaMA increased the degradation rate; pure HaMA degraded in 3 weeks, while pure GelMA in more than 5 weeks. An increase in the fraction of 2-hydroxyethylmethacrylate (HEMA) from 20% to 56% decreased the porosity and the pore size, significantly. SH-SY5Y cells migrated along the conduit in the presence of netrin-1. NeuN expression was increased in 2 weeks indicating neuronal activity.
Conclusions:
SH-SY5Y cells produced neurites in the IPN. pHEMA conduit including GelMA-HaMA IPN is a good candidate for peripheral nerve regeneration applications. As future studies, the conduit will be tested in vivo for nerve regeneration.
DOI: https://doi.org/10.37349/ebmx.2024.00005
This article belongs to the special issue Nature-Based Biomaterials for Biomedical Applications

Aim:
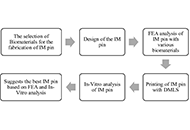
In this study, the finite elements analysis (FEA) was performed on an intramedullary (IM) pin to be used in the canine femur. The 03 different biomaterials [17-4-precipitated hardened (PH)-stainless steel (SS), nickel alloys (Ni)-625, titanium alloys (Ti)-6Al-4V] were selected for comparative FEA. In-vitro analysis was also performed in simulated body fluid (SBF) on selected biomaterials for possible application in the canine femur.
Methods:
FEA was performed on 03 different biomaterials (17-4-PH-SS, Ni-625, and Ti-6Al-4V) based on Von-Mises criteria (at an applied load of 1,500 N, cell type: tetrahedron, grit size: 0.15 mm, number of nodes: 213,989 and elements: 145,012). The distal end of the IM pin was fixed, and the load was applied to the proximal end. In-vitro analysis was performed (on a potentiostat setup) to establish the corrosion rate of various biomaterials (17-4-PH-SS, Ni-625, and Ti-6Al-4V).
Results:
The results of FEA show Ni-625 absorbed the maximum Von-Mises stress in the case of tensile and compression loading (104.12 MPa). In the case of torsion loading, the maximum Von-Mises stress was absorbed by 17-4-PH-SS (63.331 MPa). The maximum Von-Mises elastic strain (0.00093473) was observed for Ti-6Al-4V while tensile and compression and minimum deformation (0.013869 mm) in tensile loading.
Conclusions:
Based on this study, the maximum safety factor against failure (N) [ratio of 0.2% of yield strength (σy) to the Von-Mises stress (σv)] was observed as 10.75, 11.38, and 15.89, respectively, for tensile, compression, and torsional loading in the case of Ti-6Al-4V. Also, the better biocompatible material for the orthopaedic implant application based on the corrosion result is Ti-6Al-4V due to a lower corrosion rate (2.63211 × 10–10 mm/year) in comparison to 17-4-PH-SS and Ni-625. Overall, the Ti-6Al-4V is a better material than 17-4-PH-SS and Ni-625 for the intended application.
Aim:
In this study, the finite elements analysis (FEA) was performed on an intramedullary (IM) pin to be used in the canine femur. The 03 different biomaterials [17-4-precipitated hardened (PH)-stainless steel (SS), nickel alloys (Ni)-625, titanium alloys (Ti)-6Al-4V] were selected for comparative FEA. In-vitro analysis was also performed in simulated body fluid (SBF) on selected biomaterials for possible application in the canine femur.
Methods:
FEA was performed on 03 different biomaterials (17-4-PH-SS, Ni-625, and Ti-6Al-4V) based on Von-Mises criteria (at an applied load of 1,500 N, cell type: tetrahedron, grit size: 0.15 mm, number of nodes: 213,989 and elements: 145,012). The distal end of the IM pin was fixed, and the load was applied to the proximal end. In-vitro analysis was performed (on a potentiostat setup) to establish the corrosion rate of various biomaterials (17-4-PH-SS, Ni-625, and Ti-6Al-4V).
Results:
The results of FEA show Ni-625 absorbed the maximum Von-Mises stress in the case of tensile and compression loading (104.12 MPa). In the case of torsion loading, the maximum Von-Mises stress was absorbed by 17-4-PH-SS (63.331 MPa). The maximum Von-Mises elastic strain (0.00093473) was observed for Ti-6Al-4V while tensile and compression and minimum deformation (0.013869 mm) in tensile loading.
Conclusions:
Based on this study, the maximum safety factor against failure (N) [ratio of 0.2% of yield strength (σy) to the Von-Mises stress (σv)] was observed as 10.75, 11.38, and 15.89, respectively, for tensile, compression, and torsional loading in the case of Ti-6Al-4V. Also, the better biocompatible material for the orthopaedic implant application based on the corrosion result is Ti-6Al-4V due to a lower corrosion rate (2.63211 × 10–10 mm/year) in comparison to 17-4-PH-SS and Ni-625. Overall, the Ti-6Al-4V is a better material than 17-4-PH-SS and Ni-625 for the intended application.
DOI: https://doi.org/10.37349/ebmx.2024.00013
This article belongs to the special issue Metal 3D Printing of Biometals for Prostheses and Implants

Aim:
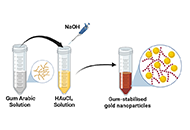
To demonstrate a simple, eco-friendly, and cost-effective green method to synthesize gold nanoparticles (AuNPs) using the aqueous extract of gum Arabic (GA) as a reducing and stabilizing agent.
Methods:
Green synthesis of nanoparticles, characterization by absorption, infra-red and fluorescence spectroscopy.
Results:
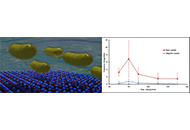
The absorption spectrum (UV-Vis) showed an absorption peak ~522 nm corresponding to the surface plasmon resonance (SPR) absorption peak of AuNPs. Transmission electron microscopy (TEM) images revealed spherical-shaped nanoparticles with an average size of 15 nm. Fourier transform infrared (FTIR) analysis showed that the nanoparticles are coated with organic compounds that are present in GA. The fluorescence quenching properties of the AuNPs were assessed by monitoring their effects on fluorescence intensity of coumarin 153 (C153) dye. The fluorescence of the dye decreased with an increase in concentration of the nanoparticles. Upon addition of the protein bovine serum albumin (BSA) to the mixture the fluorescence increased (recovery) again.
Conclusions:
The fluorescence quenching and recovery (turn-on/off system) is a valuable method for protein detection in solution. By observing the effect of BSA on the quenched fluorescence, this nanoparticle system shows promise in biomedicine, drug delivery and environmental monitoring.
Aim:
To demonstrate a simple, eco-friendly, and cost-effective green method to synthesize gold nanoparticles (AuNPs) using the aqueous extract of gum Arabic (GA) as a reducing and stabilizing agent.
Methods:
Green synthesis of nanoparticles, characterization by absorption, infra-red and fluorescence spectroscopy.
Results:
The absorption spectrum (UV-Vis) showed an absorption peak ~522 nm corresponding to the surface plasmon resonance (SPR) absorption peak of AuNPs. Transmission electron microscopy (TEM) images revealed spherical-shaped nanoparticles with an average size of 15 nm. Fourier transform infrared (FTIR) analysis showed that the nanoparticles are coated with organic compounds that are present in GA. The fluorescence quenching properties of the AuNPs were assessed by monitoring their effects on fluorescence intensity of coumarin 153 (C153) dye. The fluorescence of the dye decreased with an increase in concentration of the nanoparticles. Upon addition of the protein bovine serum albumin (BSA) to the mixture the fluorescence increased (recovery) again.
Conclusions:
The fluorescence quenching and recovery (turn-on/off system) is a valuable method for protein detection in solution. By observing the effect of BSA on the quenched fluorescence, this nanoparticle system shows promise in biomedicine, drug delivery and environmental monitoring.
DOI: https://doi.org/10.37349/ebmx.2024.00014
This article belongs to the special issue Green Nanoparticles for Biomedical Applications

Aim:
This study aimed to examine the amount of surface non-specific adsorption, or fouling, observed by Pseudomonas aeruginosa (P. aeruginosa) on a quartz crystal based acoustic wave biosensor under different flow conditions with and without an anti-fouling layer.
Methods:
An electromagnetic piezoelectric acoustic sensor (EMPAS) based on electrode free quartz crystals was used to perform the analysis. Phosphate buffered saline (PBS) was flowed over the crystal surface at various flow rates from 50 μL/min to 200 μL/min, with measurements being taken at the 43rd harmonic (~864 MHz). The crystal was either unmodified, or modified with a monoethylene glycol [2-(3-silylpropyloxy)-hydroxy-ethyl (MEG-OH)] anti-fouling layer. Overnight culture of P. aeruginosa PAO1 (PAO1) in lysogeny broth (LB) was injected into the system, and flow maintained for 30 min.
Results:
The frequency change of the EMPAS crystal after injection of bacteria into the system was found to change based on the flow rate of buffer, suggesting the flow rate has a strong effect on the level of non-specific adsorption. The MEG-OH layer drastically reduced the level of fouling observed under all flow conditions, as well as reduced the amount of variation between experiments. Flow rates of 150 μL/min or higher were found to best reduce the level of fouling observed as well as experimental variation.
Conclusions:
The MEG-OH anti-fouling layer is important for accurate and reproducible biosensing measurements due to the reduced fouling and variation during experiments. Additionally, a flow rate of 150 μL/min may prove better for measurement compared to the current standard of 50 μL/min for this type of instrument.
Aim:
This study aimed to examine the amount of surface non-specific adsorption, or fouling, observed by Pseudomonas aeruginosa (P. aeruginosa) on a quartz crystal based acoustic wave biosensor under different flow conditions with and without an anti-fouling layer.
Methods:
An electromagnetic piezoelectric acoustic sensor (EMPAS) based on electrode free quartz crystals was used to perform the analysis. Phosphate buffered saline (PBS) was flowed over the crystal surface at various flow rates from 50 μL/min to 200 μL/min, with measurements being taken at the 43rd harmonic (~864 MHz). The crystal was either unmodified, or modified with a monoethylene glycol [2-(3-silylpropyloxy)-hydroxy-ethyl (MEG-OH)] anti-fouling layer. Overnight culture of P. aeruginosa PAO1 (PAO1) in lysogeny broth (LB) was injected into the system, and flow maintained for 30 min.
Results:
The frequency change of the EMPAS crystal after injection of bacteria into the system was found to change based on the flow rate of buffer, suggesting the flow rate has a strong effect on the level of non-specific adsorption. The MEG-OH layer drastically reduced the level of fouling observed under all flow conditions, as well as reduced the amount of variation between experiments. Flow rates of 150 μL/min or higher were found to best reduce the level of fouling observed as well as experimental variation.
Conclusions:
The MEG-OH anti-fouling layer is important for accurate and reproducible biosensing measurements due to the reduced fouling and variation during experiments. Additionally, a flow rate of 150 μL/min may prove better for measurement compared to the current standard of 50 μL/min for this type of instrument.
DOI: https://doi.org/10.37349/ebmx.2023.00002

Aim:
This study aims to discover an alternative precursor with abundant source and low cost for multicolor graphene quantum dots (GQDs) preparation and application.
Methods:
In the current study, anthracite-derived multicolor GQDs were prepared at different reaction temperatures (100°–150°C), referring to the GQDs 100, GQDs 120, GQDs 130, and GQDs 150.
Results:
The GQDs 100, GQDs 120, GQDs 130, and GQDs 150 solutions were found to be orange-red, yellow-green, green, and blue under 365 nm excitation UV (ultraviolet) lamp, respectively. The X-ray photoelectron spectroscopy (XPS) data suggests high temperature intensifies oxidation of the amorphous sp3 carbon, resulting in GQDs with higher crystalline structure (Csp2). Compared with the GQDs 100 and GQDs 120, the GQDs 130 and GQDs 150 showed much better biocompatibility, which may attribute to their higher Csp2 composition and smaller size.
Conclusions:
The results suggest that GQDs 130 and GQDs 150 are ideal candidates for nanomedicine applications, e.g., drug/gene delivery and bio-imaging, etc.
Aim:
This study aims to discover an alternative precursor with abundant source and low cost for multicolor graphene quantum dots (GQDs) preparation and application.
Methods:
In the current study, anthracite-derived multicolor GQDs were prepared at different reaction temperatures (100°–150°C), referring to the GQDs 100, GQDs 120, GQDs 130, and GQDs 150.
Results:
The GQDs 100, GQDs 120, GQDs 130, and GQDs 150 solutions were found to be orange-red, yellow-green, green, and blue under 365 nm excitation UV (ultraviolet) lamp, respectively. The X-ray photoelectron spectroscopy (XPS) data suggests high temperature intensifies oxidation of the amorphous sp3 carbon, resulting in GQDs with higher crystalline structure (Csp2). Compared with the GQDs 100 and GQDs 120, the GQDs 130 and GQDs 150 showed much better biocompatibility, which may attribute to their higher Csp2 composition and smaller size.
Conclusions:
The results suggest that GQDs 130 and GQDs 150 are ideal candidates for nanomedicine applications, e.g., drug/gene delivery and bio-imaging, etc.
DOI: https://doi.org/10.37349/ebmx.2023.00003

Aim:
The pleiotropic effect of fibroblast growth factor 2 (FGF2) on promoting myogenesis, angiogenesis, and innervation makes it an ideal growth factor for treating volumetric muscle loss (VML) injuries. While an initial delivery of FGF2 has demonstrated enhanced regenerative potential, the sustained delivery of FGF2 from scaffolds with robust structural properties as well as biophysical and biochemical signaling cues has yet to be explored for treating VML. The goal of this study is to develop an instructive fibrin microthread scaffold with intrinsic topographic alignment cues as well as regenerative signaling cues and a physiologically relevant, sustained release of FGF2 to direct myogenesis and ultimately enhance functional muscle regeneration.
Methods:
Heparin was passively adsorbed or carbodiimide-conjugated to microthreads, creating a biomimetic binding strategy, mimicking FGF2 sequestration in the extracellular matrix (ECM). It was also evaluated whether FGF2 incorporated into fibrin microthreads would yield sustained release. It was hypothesized that heparin-conjugated and co-incorporated (co-inc) fibrin microthreads would facilitate sustained release of FGF2 from the scaffold and enhance in vitro myoblast proliferation and outgrowth.
Results:
Toluidine blue staining and Fourier transform infrared spectroscopy confirmed that carbodiimide-conjugated heparin bound to fibrin microthreads in a dose-dependent manner. Release kinetics revealed that heparin-conjugated fibrin microthreads exhibited sustained release of FGF2 over a period of one week. An in vitro assay demonstrated that FGF2 released from microthreads remained bioactive, stimulating myoblast proliferation over four days. Finally, a cellular outgrowth assay suggests that FGF2 promotes increased outgrowth onto microthreads.
Conclusions:
It was anticipated that the combined effects of fibrin microthread structural properties, topographic alignment cues, and FGF2 release profiles will facilitate the fabrication of a biomimetic scaffold that enhances the regeneration of functional muscle tissue for the treatment of VML injuries.
Aim:
The pleiotropic effect of fibroblast growth factor 2 (FGF2) on promoting myogenesis, angiogenesis, and innervation makes it an ideal growth factor for treating volumetric muscle loss (VML) injuries. While an initial delivery of FGF2 has demonstrated enhanced regenerative potential, the sustained delivery of FGF2 from scaffolds with robust structural properties as well as biophysical and biochemical signaling cues has yet to be explored for treating VML. The goal of this study is to develop an instructive fibrin microthread scaffold with intrinsic topographic alignment cues as well as regenerative signaling cues and a physiologically relevant, sustained release of FGF2 to direct myogenesis and ultimately enhance functional muscle regeneration.
Methods:
Heparin was passively adsorbed or carbodiimide-conjugated to microthreads, creating a biomimetic binding strategy, mimicking FGF2 sequestration in the extracellular matrix (ECM). It was also evaluated whether FGF2 incorporated into fibrin microthreads would yield sustained release. It was hypothesized that heparin-conjugated and co-incorporated (co-inc) fibrin microthreads would facilitate sustained release of FGF2 from the scaffold and enhance in vitro myoblast proliferation and outgrowth.
Results:
Toluidine blue staining and Fourier transform infrared spectroscopy confirmed that carbodiimide-conjugated heparin bound to fibrin microthreads in a dose-dependent manner. Release kinetics revealed that heparin-conjugated fibrin microthreads exhibited sustained release of FGF2 over a period of one week. An in vitro assay demonstrated that FGF2 released from microthreads remained bioactive, stimulating myoblast proliferation over four days. Finally, a cellular outgrowth assay suggests that FGF2 promotes increased outgrowth onto microthreads.
Conclusions:
It was anticipated that the combined effects of fibrin microthread structural properties, topographic alignment cues, and FGF2 release profiles will facilitate the fabrication of a biomimetic scaffold that enhances the regeneration of functional muscle tissue for the treatment of VML injuries.
DOI: https://doi.org/10.37349/ebmx.2024.00006

Aim:
Since decades, decellularized extracellular matrix (dECM)-derived materials have received worldwide attention as promising biomaterials for tissue engineering and biomedical applications. Soluble dECM is a versatile raw material that can be easily engineered into the desired shapes and structures. However, there are still some limitations restricting its use, including low hydrophilicity and smooth surfaces, which negatively influence cell adhesion/spreading. The objective of the present study was to investigate surface modification by nitrogen/hydrogen (N2/H2) low-pressure cold plasma treatment as a potential technique to improve the biological response of bovine pericardium dECM films.
Methods:
Bovine pericardium dECM was enzymatically digested and lyophilized prior to the preparation of thin films via solvent-casting method. Changes in surface properties after plasma treatment were investigated using water contact angle (WCA) and X-ray photoelectron spectroscopy (XPS) measurements. Immunofluorescence staining and resazurin assay for human dermal fibroblasts (HDFs) cultured on the dECM films were used to assess the bioactivity of dECM films. Finally, the hemocompatibility of the films was investigated via clotting time and hemolysis assay.
Results:
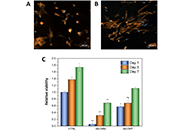
WCA and XPS results revealed that oxygen (O)- and N-containing functional groups were incorporated onto the film surface and an increase in hydrophilicity was observed after plasma treatment. In vitro experiments showed that cell adhesion in plasma-treated dECM films is much faster if compared to the untreated controls. Moreover, the fibroblast proliferation increased after plasma surface modifications. Finally, the hemocompatibility analysis results indicated a delayed blood clotting and no hemolytic effects for all the tested samples.
Conclusions:
These findings confirmed the potential of dECM as raw material for biocompatible thin films fabrication. Additionally, plasma surface treatment emerged as an eco-friendly and cost-effective strategy to enhance in vitro cell attachment and proliferation on dECM films, expanding their applications in biomedicine.
Aim:
Since decades, decellularized extracellular matrix (dECM)-derived materials have received worldwide attention as promising biomaterials for tissue engineering and biomedical applications. Soluble dECM is a versatile raw material that can be easily engineered into the desired shapes and structures. However, there are still some limitations restricting its use, including low hydrophilicity and smooth surfaces, which negatively influence cell adhesion/spreading. The objective of the present study was to investigate surface modification by nitrogen/hydrogen (N2/H2) low-pressure cold plasma treatment as a potential technique to improve the biological response of bovine pericardium dECM films.
Methods:
Bovine pericardium dECM was enzymatically digested and lyophilized prior to the preparation of thin films via solvent-casting method. Changes in surface properties after plasma treatment were investigated using water contact angle (WCA) and X-ray photoelectron spectroscopy (XPS) measurements. Immunofluorescence staining and resazurin assay for human dermal fibroblasts (HDFs) cultured on the dECM films were used to assess the bioactivity of dECM films. Finally, the hemocompatibility of the films was investigated via clotting time and hemolysis assay.
Results:
WCA and XPS results revealed that oxygen (O)- and N-containing functional groups were incorporated onto the film surface and an increase in hydrophilicity was observed after plasma treatment. In vitro experiments showed that cell adhesion in plasma-treated dECM films is much faster if compared to the untreated controls. Moreover, the fibroblast proliferation increased after plasma surface modifications. Finally, the hemocompatibility analysis results indicated a delayed blood clotting and no hemolytic effects for all the tested samples.
Conclusions:
These findings confirmed the potential of dECM as raw material for biocompatible thin films fabrication. Additionally, plasma surface treatment emerged as an eco-friendly and cost-effective strategy to enhance in vitro cell attachment and proliferation on dECM films, expanding their applications in biomedicine.
DOI: https://doi.org/10.37349/ebmx.2024.00007

Neurodegenerative diseases (NDDs) gradually affect neurons impacting both their function and structure, and they afflict millions worldwide. Detecting these conditions before symptoms arise is crucial for better prognosis and duality of life, given that the disease processes often begin years earlier. Yet, reliable and affordable methods to diagnose NDDs in these stages are currently lacking. There’s a growing interest in using circulating extracellular vesicles (EVs), like small EVs (sEVs) also known as exosomes, as potential sources of markers for screening, diagnosing, and monitoring NDDs. This interest stems from evidence showing that these EVs can carry brain pathological proteins implicated in NDD pathology, and they can even traverse the blood-brain barrier. This review focuses on the creation of EVs, particularly sEVs with a size of less than 200 nanometers, methods for isolating sEVs, and recent advancements in biosensor development to detect NDD-related markers found in sEVs. Furthermore, it explores the potential of sEVs in diagnosing four major NDDs: Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), and multiple system atrophy (MSA).
Neurodegenerative diseases (NDDs) gradually affect neurons impacting both their function and structure, and they afflict millions worldwide. Detecting these conditions before symptoms arise is crucial for better prognosis and duality of life, given that the disease processes often begin years earlier. Yet, reliable and affordable methods to diagnose NDDs in these stages are currently lacking. There’s a growing interest in using circulating extracellular vesicles (EVs), like small EVs (sEVs) also known as exosomes, as potential sources of markers for screening, diagnosing, and monitoring NDDs. This interest stems from evidence showing that these EVs can carry brain pathological proteins implicated in NDD pathology, and they can even traverse the blood-brain barrier. This review focuses on the creation of EVs, particularly sEVs with a size of less than 200 nanometers, methods for isolating sEVs, and recent advancements in biosensor development to detect NDD-related markers found in sEVs. Furthermore, it explores the potential of sEVs in diagnosing four major NDDs: Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), and multiple system atrophy (MSA).
DOI: https://doi.org/10.37349/ebmx.2024.00008

Nanomedicine, a convergence of nanotechnology and medical sciences, has unleashed transformative potential in healthcare. However, harnessing the benefits of nanomedicine requires a thorough understanding of its regulatory landscape. An in-depth discussion of regulatory considerations, including molecular safety assessment, harmonization of the regulatory landscape, and shaping the future of innovation, is presented in this discourse. The molecular safety assessment entails evaluating interactions between nanoparticles and biomolecules, ensuring compatibility at the molecular level. Harmonization involves developing international standards and guidelines for a consistent regulatory approach, while shaping innovations emphasizes integrating molecular safety assessments into early stages of development. Challenges encompass the need for standardized assessment methods, balancing innovation with safety, and addressing unique features of novel molecular designs. As the nanomedicine landscape evolves, effective regulatory strategies must navigate the intricate interplay of molecules and technologies, ensuring both patient access and product safety.
Nanomedicine, a convergence of nanotechnology and medical sciences, has unleashed transformative potential in healthcare. However, harnessing the benefits of nanomedicine requires a thorough understanding of its regulatory landscape. An in-depth discussion of regulatory considerations, including molecular safety assessment, harmonization of the regulatory landscape, and shaping the future of innovation, is presented in this discourse. The molecular safety assessment entails evaluating interactions between nanoparticles and biomolecules, ensuring compatibility at the molecular level. Harmonization involves developing international standards and guidelines for a consistent regulatory approach, while shaping innovations emphasizes integrating molecular safety assessments into early stages of development. Challenges encompass the need for standardized assessment methods, balancing innovation with safety, and addressing unique features of novel molecular designs. As the nanomedicine landscape evolves, effective regulatory strategies must navigate the intricate interplay of molecules and technologies, ensuring both patient access and product safety.
DOI: https://doi.org/10.37349/ebmx.2024.00009

Aim:
Polymethylmethacrylate bone cement is often used to reconstruct critical-sized defects generated by the surgical resection of spinal metastases. Residual tumor cells after a resection can drive recurrence and destabilization. Doxorubicin (DOX) is a common chemotherapeutic drug with unwanted side-effects when administered systemically. Mesoporous silica nanoparticles (NPs) are gaining attention for targeted drug delivery to bypass the negative side effects associated with systemic drug administration. An NP-functionalized cement was developed for the local release of DOX and its ability to suppress cancer cells was tested.
Methods:
DOX was loaded onto NPs which were then mixed into the cement. Static contact angles were measured. Drug release profiles were obtained over a period of 4 weeks. Cement constructs were incubated with two-dimensional (2D) cultures of human bone-marrow derived mesenchymal stem cells and human osteoblasts, as well as 2D and three-dimensional (3D) cultures of breast and prostate cancer cell lines. Cell metabolic activity and viability were evaluated. Cell migration and spheroid growth of cancer cell lines were assessed in collagen-coated spheroid cultures.
Results:
NPs were homogenously dispersed and did not alter the mechanical strength nor the wettability of the cement. A sustained DOX release profile was achieved with the addition of NPs to the bone cement. The release profile of DOX from NP cement may be modified by varying the amount of the drug loaded onto the NPs and the proportion of NPs in the cement. Cancer cells treated with the cement constructs showed a dose- and time-dependent inhibition, with minimal toxicity against healthy cells. Cancer cell migration and spheroid growth were impaired in 3D culture.
Conclusions:
NPs were shown to be essential for sustained DOX release from bone cement. DOX-loaded NP cement can inhibit cancer cells and impair their migration, with strong potential for in vivo translation studies.
Aim:
Polymethylmethacrylate bone cement is often used to reconstruct critical-sized defects generated by the surgical resection of spinal metastases. Residual tumor cells after a resection can drive recurrence and destabilization. Doxorubicin (DOX) is a common chemotherapeutic drug with unwanted side-effects when administered systemically. Mesoporous silica nanoparticles (NPs) are gaining attention for targeted drug delivery to bypass the negative side effects associated with systemic drug administration. An NP-functionalized cement was developed for the local release of DOX and its ability to suppress cancer cells was tested.
Methods:
DOX was loaded onto NPs which were then mixed into the cement. Static contact angles were measured. Drug release profiles were obtained over a period of 4 weeks. Cement constructs were incubated with two-dimensional (2D) cultures of human bone-marrow derived mesenchymal stem cells and human osteoblasts, as well as 2D and three-dimensional (3D) cultures of breast and prostate cancer cell lines. Cell metabolic activity and viability were evaluated. Cell migration and spheroid growth of cancer cell lines were assessed in collagen-coated spheroid cultures.
Results:
NPs were homogenously dispersed and did not alter the mechanical strength nor the wettability of the cement. A sustained DOX release profile was achieved with the addition of NPs to the bone cement. The release profile of DOX from NP cement may be modified by varying the amount of the drug loaded onto the NPs and the proportion of NPs in the cement. Cancer cells treated with the cement constructs showed a dose- and time-dependent inhibition, with minimal toxicity against healthy cells. Cancer cell migration and spheroid growth were impaired in 3D culture.
Conclusions:
NPs were shown to be essential for sustained DOX release from bone cement. DOX-loaded NP cement can inhibit cancer cells and impair their migration, with strong potential for in vivo translation studies.
DOI: https://doi.org/10.37349/ebmx.2024.00010

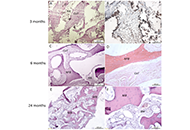
The use of autologous cartilage and bone grafts remains the gold standard in augmentation rhinoplasty performed to reconstruct of the nasal dorsum. Meanwhile, limited number of available sources, donor site morbidity, and unpredictable graft resorption represent significant disadvantages of autografting. The aim of this study is to test combination of autologous stromal vascular fraction (SVF) and commercially available bone substitutes (BSs) as new tissue-engineered grafting material (GM) for rhinoplasty. A series of consecutive cases includes four adult patients who underwent rhinoplasty to correct saddle nose deformity (SND) using the new graft technique. SVF was isolated from liposuction aspirate using standard methodology of enzymatic digestion. Two types of BSs were combined with SVF: Bio-Oss granules to create a moldable graft (M-graft), and block-shaped BoneMedik-S to create rigid grafts (R-grafts). The moderate SND was treated using an M-graft. In case of major or complex SND, the nasal dorsum was reconstructed with dorso-columellar L-shaped framework made of R-grafts. The results were evaluated over a period of 6 months to 3 years postoperatively using photogrammetry and FACE-Q appearance appraisal scales. Computerised tomography (CT) scanning of the reconstructed nose and histological analysis of grafted material were also carried out. No complications were observed. The photograms show the restoration of the correct contour of the nose. FACE-Q appraisal scale scores increased significantly, including satisfaction with nose appearance, psychological well-being, and social function. In CT evaluation, there was no substantial resorption or warping of the grafts. Histological findings show osteogenic remodeling of the grafted material. Thus, combining autologous SVF with BSs is a promising strategy for developing rhinoplasty GM.
The use of autologous cartilage and bone grafts remains the gold standard in augmentation rhinoplasty performed to reconstruct of the nasal dorsum. Meanwhile, limited number of available sources, donor site morbidity, and unpredictable graft resorption represent significant disadvantages of autografting. The aim of this study is to test combination of autologous stromal vascular fraction (SVF) and commercially available bone substitutes (BSs) as new tissue-engineered grafting material (GM) for rhinoplasty. A series of consecutive cases includes four adult patients who underwent rhinoplasty to correct saddle nose deformity (SND) using the new graft technique. SVF was isolated from liposuction aspirate using standard methodology of enzymatic digestion. Two types of BSs were combined with SVF: Bio-Oss granules to create a moldable graft (M-graft), and block-shaped BoneMedik-S to create rigid grafts (R-grafts). The moderate SND was treated using an M-graft. In case of major or complex SND, the nasal dorsum was reconstructed with dorso-columellar L-shaped framework made of R-grafts. The results were evaluated over a period of 6 months to 3 years postoperatively using photogrammetry and FACE-Q appearance appraisal scales. Computerised tomography (CT) scanning of the reconstructed nose and histological analysis of grafted material were also carried out. No complications were observed. The photograms show the restoration of the correct contour of the nose. FACE-Q appraisal scale scores increased significantly, including satisfaction with nose appearance, psychological well-being, and social function. In CT evaluation, there was no substantial resorption or warping of the grafts. Histological findings show osteogenic remodeling of the grafted material. Thus, combining autologous SVF with BSs is a promising strategy for developing rhinoplasty GM.
DOI: https://doi.org/10.37349/ebmx.2024.00011
This article belongs to the special issue Bioinspired Material for Regenerative Medicine

DOI: https://doi.org/10.37349/ebmx.2024.00012
This article belongs to the special issue Innovations in Biomaterials for Dentistry and Oral Surgery

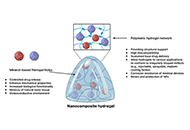
Mineral nanoparticles and osteoinductive biomaterials are essential in advancing bone regeneration by addressing skeletal conditions and injuries that compromise structural integrity and functionality. These biomaterials stimulate the differentiation of precursor cells into osteoblasts, creating biocompatible environments conducive to bone tissue regeneration. Among the most promising innovations, mineral-based nanoparticles and nanocomposite hydrogels have emerged as effective strategies for enhancing osteoinductive potential. This review explores the diverse types of osteoinductive biomaterials, including natural sources, synthetic compounds, and hybrid designs that incorporate mineralized nanoparticles. Emphasis is placed on polymeric hydrogels as delivery platforms for these materials, highlighting their dual role as structural supports and bioactive agents that promote osteogenesis. Challenges such as immune rejection, biodegradability, mechanical stability, and short in vivo residence time are critically discussed, alongside their impact on clinical translation. By presenting a comprehensive analysis of mechanisms, applications, and limitations, this review identifies opportunities for integrating osteoinductive biomaterials with emerging fields like immunology and biomechanics. Ultimately, this work aims to provide actionable insights and advance the development of novel, clinically relevant solutions that improve patient outcomes and address the growing global need for effective bone repair and regeneration.
Mineral nanoparticles and osteoinductive biomaterials are essential in advancing bone regeneration by addressing skeletal conditions and injuries that compromise structural integrity and functionality. These biomaterials stimulate the differentiation of precursor cells into osteoblasts, creating biocompatible environments conducive to bone tissue regeneration. Among the most promising innovations, mineral-based nanoparticles and nanocomposite hydrogels have emerged as effective strategies for enhancing osteoinductive potential. This review explores the diverse types of osteoinductive biomaterials, including natural sources, synthetic compounds, and hybrid designs that incorporate mineralized nanoparticles. Emphasis is placed on polymeric hydrogels as delivery platforms for these materials, highlighting their dual role as structural supports and bioactive agents that promote osteogenesis. Challenges such as immune rejection, biodegradability, mechanical stability, and short in vivo residence time are critically discussed, alongside their impact on clinical translation. By presenting a comprehensive analysis of mechanisms, applications, and limitations, this review identifies opportunities for integrating osteoinductive biomaterials with emerging fields like immunology and biomechanics. Ultimately, this work aims to provide actionable insights and advance the development of novel, clinically relevant solutions that improve patient outcomes and address the growing global need for effective bone repair and regeneration.
DOI: https://doi.org/10.37349/ebmx.2025.101332

Aim:
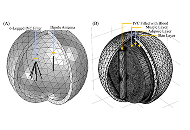
To show that a wireless-powered thrombolytic filter can be used in the treatment of venous thromboembolism (VTE) as an alternative to the existing VTE therapies, which have serious side effects.
Methods:
The wireless-powered thrombolytic filter that we propose combines the positive attributes of anticoagulants and thrombolytics, through the capture and dissolution of blood clots, without the associated adverse effects of existing treatments. The filter absorbs radio-frequency energy from a source and converts it into heat at the thrombolytic filter.
Results:
We used computer simulations with COMSOL and lab experiments to demonstrate that a wireless-powered thrombolytic filter can be heated up through the absorption of radio-frequency energy from an external source.
Conclusions:
We demonstrate that a wireless-powered thrombolytic filter has the potential to be used in the treatment of VTE, since it can be designed to absorb energy from an external radio-frequency source and convert it to heat that is sufficient to dissolve blood clots captured by the thrombolytic filter.
Aim:
To show that a wireless-powered thrombolytic filter can be used in the treatment of venous thromboembolism (VTE) as an alternative to the existing VTE therapies, which have serious side effects.
Methods:
The wireless-powered thrombolytic filter that we propose combines the positive attributes of anticoagulants and thrombolytics, through the capture and dissolution of blood clots, without the associated adverse effects of existing treatments. The filter absorbs radio-frequency energy from a source and converts it into heat at the thrombolytic filter.
Results:
We used computer simulations with COMSOL and lab experiments to demonstrate that a wireless-powered thrombolytic filter can be heated up through the absorption of radio-frequency energy from an external source.
Conclusions:
We demonstrate that a wireless-powered thrombolytic filter has the potential to be used in the treatment of VTE, since it can be designed to absorb energy from an external radio-frequency source and convert it to heat that is sufficient to dissolve blood clots captured by the thrombolytic filter.
DOI: https://doi.org/10.37349/ebmx.2025.101333
This article belongs to the special issue Trends in Biomaterials Research for Cardiovascular Applications

Aim:
The purpose of this study is to investigate the deformability and strength of silicone breast implant shells from different manufactures as a function of implantation time.
Methods:
The strength properties of about 200 shells of Eurosilicone, Mentor, Motiva, Allergan, Arion, PIP silicone breast implants removed for various reasons during repeated surgeries with a period of stay in the body from 6 months to 29 years were measured and compared with the corresponding properties of four shells of unused Eurosilicone and Motiva implants. Deformation was measured using a videoXtens extensometer.
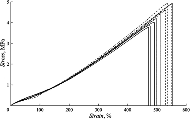
Results:
The mechanical properties of the Allergan implant shell are almost completely consistent with the properties of the Eurosilicone shell after 9 years of use. The Mentor implants showed greater strength and stiffness. The Motiva implant shells initially had higher ultimate properties—rupture stress and rupture strain—in comparison with the Eurosilicone implant shells.
Conclusions:
The strength and deformation properties of all examined breast implant shells decrease in the course of time. After 13 years of use, the strength of breast implants is halved and their rupture strain is reduced by one third. The main mechanism responsible for loss of strength is the accumulation of microdamages during long-term use of breast implants. The thickness of the nano-textured shells of Motiva implants is twice less of the Eurosilicone implant shell thickness. This was possible due to a significant increase in the mechanical properties of Motiva shells.
Aim:
The purpose of this study is to investigate the deformability and strength of silicone breast implant shells from different manufactures as a function of implantation time.
Methods:
The strength properties of about 200 shells of Eurosilicone, Mentor, Motiva, Allergan, Arion, PIP silicone breast implants removed for various reasons during repeated surgeries with a period of stay in the body from 6 months to 29 years were measured and compared with the corresponding properties of four shells of unused Eurosilicone and Motiva implants. Deformation was measured using a videoXtens extensometer.
Results:
The mechanical properties of the Allergan implant shell are almost completely consistent with the properties of the Eurosilicone shell after 9 years of use. The Mentor implants showed greater strength and stiffness. The Motiva implant shells initially had higher ultimate properties—rupture stress and rupture strain—in comparison with the Eurosilicone implant shells.
Conclusions:
The strength and deformation properties of all examined breast implant shells decrease in the course of time. After 13 years of use, the strength of breast implants is halved and their rupture strain is reduced by one third. The main mechanism responsible for loss of strength is the accumulation of microdamages during long-term use of breast implants. The thickness of the nano-textured shells of Motiva implants is twice less of the Eurosilicone implant shell thickness. This was possible due to a significant increase in the mechanical properties of Motiva shells.
DOI: https://doi.org/10.37349/ebmx.2025.101334
This article belongs to the special issue The Use of 2D Materials in Bio-tribology

Aim:
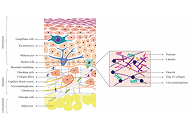
This study evaluated the impact of retinal extracellular matrix (ECM) and key biomaterial substrates on the motility of transplantable retinal cells with genomic manipulation, using the therapeutic molecule, Topoisomerase II beta (Top2b), as a model.
Methods:
Tests first applied in ovo electroporation to examine the effects of a pharmacological Top2b inhibitor (ICRF-193) on progenitor motility and development of embryonic retina. Complementary qRT-PCR tests measured changes in select cadherin molecules in response to treatment. In vitro transfection produced cultured retinal progenitor cell groups with Top2b overexpression and Top2b knockdown. Differences in the adhesion and motility of Top2b altered groups, compared to wildtype cells, were measured upon biomaterial substrates used in emerging transplantation matrixes.
Results:
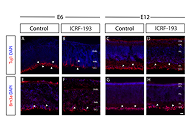
Data illustrated significant differences in the number and spacing of retinal ganglion cells when retina was treated with ICRF-193, as well as downregulation of several key cadherin molecules. Cultured retinal progenitors with Top2b knockdown and Top2b overexpression exhibited different expression of chemotactic receptors, adhesion parameters, and modalities of migration upon substrates of laminin, poly-L-lysine, and collagen IV. Significant changes in cell morphology and surface area were also measured compared to wildtype cells.
Conclusions:
Corroborating in vivo and in vitro data support Top2b as a therapeutic target for retinal progenitor motility but indicate significant differences in the migration of Top2b altered cells upon substrates used in transplantation. These data highlight the therapeutic advantages of bioinspired materials developed to aid the motility of replacement cells with modified genetic expression to improve transplantation outcomes across the nervous system.
Aim:
This study evaluated the impact of retinal extracellular matrix (ECM) and key biomaterial substrates on the motility of transplantable retinal cells with genomic manipulation, using the therapeutic molecule, Topoisomerase II beta (Top2b), as a model.
Methods:
Tests first applied in ovo electroporation to examine the effects of a pharmacological Top2b inhibitor (ICRF-193) on progenitor motility and development of embryonic retina. Complementary qRT-PCR tests measured changes in select cadherin molecules in response to treatment. In vitro transfection produced cultured retinal progenitor cell groups with Top2b overexpression and Top2b knockdown. Differences in the adhesion and motility of Top2b altered groups, compared to wildtype cells, were measured upon biomaterial substrates used in emerging transplantation matrixes.
Results:
Data illustrated significant differences in the number and spacing of retinal ganglion cells when retina was treated with ICRF-193, as well as downregulation of several key cadherin molecules. Cultured retinal progenitors with Top2b knockdown and Top2b overexpression exhibited different expression of chemotactic receptors, adhesion parameters, and modalities of migration upon substrates of laminin, poly-L-lysine, and collagen IV. Significant changes in cell morphology and surface area were also measured compared to wildtype cells.
Conclusions:
Corroborating in vivo and in vitro data support Top2b as a therapeutic target for retinal progenitor motility but indicate significant differences in the migration of Top2b altered cells upon substrates used in transplantation. These data highlight the therapeutic advantages of bioinspired materials developed to aid the motility of replacement cells with modified genetic expression to improve transplantation outcomes across the nervous system.
DOI: https://doi.org/10.37349/ebmx.2025.101335
This article belongs to the special issue Bioinspired Material for Regenerative Medicine

This review highlights the challenges of current wound healing methods, such as scar formation and limited regeneration, and emphasizes the potential of tissue engineering to address these issues. Chitosan, a biopolymer derived from chitin, has garnered significant attention in epidermal-dermal wound healing due to its exceptional biocompatibility, biodegradability, and versatile functional properties. This review article delves into the diverse roles of chitosan, with a particular focus on its use as a scaffold material with fine-tunable physicochemical and biological properties for accelerated wound healing. While bare chitosan provides a suitable microenvironment for cell adhesion and proliferation, it exhibits limited mechanical strength and drug-delivery properties. However, combining it with other natural and synthetic polymers and nanoparticles facilitates drug and biosignal delivery and enhances biocompatibility and antibacterial activity. Furthermore, the review covers various chemical modifications of chitosan, including quaternization and methacrylation, to improve biocompatibility, water solubility and mechanical strength, for developing advanced wound dressings for effective skin regeneration. The review also discusses various types of smart chitosan hydrogels and the clinical translation of chitosan based scaffolds for wound healing and tissue regeneration applications. Finally, it discusses the integration of 3D bioprinting techniques for creating complex, cell-incorporated scaffolds for advanced wound healing therapies.
This review highlights the challenges of current wound healing methods, such as scar formation and limited regeneration, and emphasizes the potential of tissue engineering to address these issues. Chitosan, a biopolymer derived from chitin, has garnered significant attention in epidermal-dermal wound healing due to its exceptional biocompatibility, biodegradability, and versatile functional properties. This review article delves into the diverse roles of chitosan, with a particular focus on its use as a scaffold material with fine-tunable physicochemical and biological properties for accelerated wound healing. While bare chitosan provides a suitable microenvironment for cell adhesion and proliferation, it exhibits limited mechanical strength and drug-delivery properties. However, combining it with other natural and synthetic polymers and nanoparticles facilitates drug and biosignal delivery and enhances biocompatibility and antibacterial activity. Furthermore, the review covers various chemical modifications of chitosan, including quaternization and methacrylation, to improve biocompatibility, water solubility and mechanical strength, for developing advanced wound dressings for effective skin regeneration. The review also discusses various types of smart chitosan hydrogels and the clinical translation of chitosan based scaffolds for wound healing and tissue regeneration applications. Finally, it discusses the integration of 3D bioprinting techniques for creating complex, cell-incorporated scaffolds for advanced wound healing therapies.
DOI: https://doi.org/10.37349/ebmx.2025.101336
This article belongs to the special issue Nature-Based Biomaterials for Biomedical Applications

Aim:
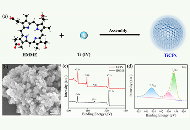
Multifunctional nanomaterials with photodynamic-sonodynamic therapy (PSDT) potential offer significant advantages in cancer treatment. However, designing and preparing single-component “two-in-one” multifunctional nanomaterials remains challenging. Hematoporphyrin monomethyl ether (HMME), a second-generation porphyrin-related sonosensitizer, is a porphyrin derivative with two asymmetric carboxyl groups. Notably, the carboxyl groups in HMME can coordinate with metal ions to construct metal-organic coordination nanomaterials (MCPs). Titanium (Ti), a biocompatible metal element, is commonly used in medical devices such as implantable metal alloys. Therefore, this study reported the synthesis of “two-in-one” type Ti-HMME coordination nanomaterials (TiCPs) as efficient nanoscale photo/sonosensitizers.
Methods:
Under a nitrogen atmosphere, TiCPs were synthesized via self-assembly between HMME and Ti4+ ions.
Results:
The average particle size of TiCPs was approximately 70 nm. Additionally, TiCPs contained the photo/sonosensitizer HMME, which could convert O2 into cytotoxic reactive oxygen species (ROS) under light and ultrasound (US) excitation. The generation of ROS could be detected using 1,3-diphenylisobenzofuran (DPBF). When the mixed solution (TiCPs + DPBF) was irradiated with light, the DPBF peak rapidly decreased with increasing irradiation time, indicating the production of ROS by TiCPs under light. Similarly, the absorbance of TiCPs + DPBF significantly decreased with increasing US time, demonstrating the sonodynamic effect of TiCPs + US. After 10 min of light or US excitation, 49.4% (Light) and 38.1% (US) of DPBF were oxidized by ROS generated by TiCPs, showcasing excellent photodynamic/sonodynamic effects. In vitro cell experiments further demonstrated that TiCPs had excellent biocompatibility, could be effectively internalized by cells, and significantly reduced cell viability under light and US excitation, effectively killing tumor cells.
Conclusions:
This study not only demonstrated TiCPs as “two-in-one” type multifunctional nanomaterials for PSDT but also provided insights into designing other photo/sonosensitizer molecules with similar HMME structures for tumor theranostics.
Aim:
Multifunctional nanomaterials with photodynamic-sonodynamic therapy (PSDT) potential offer significant advantages in cancer treatment. However, designing and preparing single-component “two-in-one” multifunctional nanomaterials remains challenging. Hematoporphyrin monomethyl ether (HMME), a second-generation porphyrin-related sonosensitizer, is a porphyrin derivative with two asymmetric carboxyl groups. Notably, the carboxyl groups in HMME can coordinate with metal ions to construct metal-organic coordination nanomaterials (MCPs). Titanium (Ti), a biocompatible metal element, is commonly used in medical devices such as implantable metal alloys. Therefore, this study reported the synthesis of “two-in-one” type Ti-HMME coordination nanomaterials (TiCPs) as efficient nanoscale photo/sonosensitizers.
Methods:
Under a nitrogen atmosphere, TiCPs were synthesized via self-assembly between HMME and Ti4+ ions.
Results:
The average particle size of TiCPs was approximately 70 nm. Additionally, TiCPs contained the photo/sonosensitizer HMME, which could convert O2 into cytotoxic reactive oxygen species (ROS) under light and ultrasound (US) excitation. The generation of ROS could be detected using 1,3-diphenylisobenzofuran (DPBF). When the mixed solution (TiCPs + DPBF) was irradiated with light, the DPBF peak rapidly decreased with increasing irradiation time, indicating the production of ROS by TiCPs under light. Similarly, the absorbance of TiCPs + DPBF significantly decreased with increasing US time, demonstrating the sonodynamic effect of TiCPs + US. After 10 min of light or US excitation, 49.4% (Light) and 38.1% (US) of DPBF were oxidized by ROS generated by TiCPs, showcasing excellent photodynamic/sonodynamic effects. In vitro cell experiments further demonstrated that TiCPs had excellent biocompatibility, could be effectively internalized by cells, and significantly reduced cell viability under light and US excitation, effectively killing tumor cells.
Conclusions:
This study not only demonstrated TiCPs as “two-in-one” type multifunctional nanomaterials for PSDT but also provided insights into designing other photo/sonosensitizer molecules with similar HMME structures for tumor theranostics.
DOI: https://doi.org/10.37349/ebmx.2025.101337

