



DOI: https://doi.org/10.37349/ec.2023.00001

Lipoprotein(a) [Lp(a)] is composed of a low-density lipoprotein (LDL) and glycoprotein (a)—apolipoprotein(a) [apo(a)]. The size and concentration of Lp(a) in serum can vary among individuals and is determined by genetic factors. The environmental factors, diet, and physical activity have a negligible effect on Lp(a) level. Observational, epidemiological, and genetic studies improved that high levels of Lp(a) > 50 mg/dL (> 125 nmol/L) have been associated with an increased risk of myocardial infarction (MI), stroke, and calcific aortic valve stenosis (CAVS). It is recommended to measure Lp(a) at least once in adults to identify individuals with a high cardiovascular risk. This screening is particularly important in certain populations, including: youth with a history of ischemic stroke or a family history of premature atherosclerotic cardiovascular disease (CVD; ASCVD) or high Lp(a), individuals with recurrent cardiovascular events despite optimal hypolipemic treatment and no other identifiable risk factors or patients with familial hypercholesterolemia (FH). Considering Lp(a) levels in the evaluation of cardiovascular risk can provide valuable information for risk stratification and management decisions. However, it’s important to note that the treatments of elevated level of Lp(a) are limited. In recent years, there has been ongoing research and development of new drugs targeting Lp(a): pelacarsen—antisense oligonucleotide (ASO), and olpasiran—a small interfering RNA (siRNA).
Lipoprotein(a) [Lp(a)] is composed of a low-density lipoprotein (LDL) and glycoprotein (a)—apolipoprotein(a) [apo(a)]. The size and concentration of Lp(a) in serum can vary among individuals and is determined by genetic factors. The environmental factors, diet, and physical activity have a negligible effect on Lp(a) level. Observational, epidemiological, and genetic studies improved that high levels of Lp(a) > 50 mg/dL (> 125 nmol/L) have been associated with an increased risk of myocardial infarction (MI), stroke, and calcific aortic valve stenosis (CAVS). It is recommended to measure Lp(a) at least once in adults to identify individuals with a high cardiovascular risk. This screening is particularly important in certain populations, including: youth with a history of ischemic stroke or a family history of premature atherosclerotic cardiovascular disease (CVD; ASCVD) or high Lp(a), individuals with recurrent cardiovascular events despite optimal hypolipemic treatment and no other identifiable risk factors or patients with familial hypercholesterolemia (FH). Considering Lp(a) levels in the evaluation of cardiovascular risk can provide valuable information for risk stratification and management decisions. However, it’s important to note that the treatments of elevated level of Lp(a) are limited. In recent years, there has been ongoing research and development of new drugs targeting Lp(a): pelacarsen—antisense oligonucleotide (ASO), and olpasiran—a small interfering RNA (siRNA).
DOI: https://doi.org/10.37349/ec.2023.00015
This article belongs to the special issue Common cardiovascular target for a wide gamut of contemporary health problems – thrombotic and arrhythmic sides of an inflammatory coin

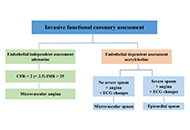
Coronary vasospasm stands as a widely acknowledged and frequent culprit behind chest pain, acute coronary syndrome, and sudden cardiac death, yet it remains a challenging diagnosis. Current guidelines recommend invasive coronary function testing to assess pathophysiology and mechanisms and to define treatment. In reality, this protocol is rarely applied, because it necessitates extended occupation of the cath lab, repetitive administration of nephrotoxic iodine contrast agents, the need for repeated testing on both coronary arteries leading to considerable radiation exposure, and significant direct expenses. The promising perspective for vasospasm testing is a noninvasive approach with advanced echocardiographic techniques, such as transthoracic Doppler echocardiography, with more sensitive indicators of ischemia. Hyperventilation and exercise tests are used for vasospasm directed testing, with assessment of the new parameters: coronary flow velocities and reserve, allowing to see deeper into macro and microvascular pathophysiology. Association between coronary flow, global longitudinal strain and microvascular dysfunction (MVD) and impaired values at hyperemia was previously demonstrated. Reduction in coronary flow velocity (CFV) despite heightened myocardial oxygen consumption and double product during hyperventilation are indicative of coronary vasospasm. Normal coronary angiography finding in patients with documented evidence of ischemia should initiate additional diagnostic testing in order to increase the yield of specific diagnosis in patients with suspected vasospasm, which could help to personalize treatment and prognosis. In order to achieve this, non-invasive provocative stress echocardiography tests should be included in the diagnostic workup. This approach, characterized by its simplicity, feasibility, safety, and efficacy, is currently undergoing extensive testing on a large scale.
Coronary vasospasm stands as a widely acknowledged and frequent culprit behind chest pain, acute coronary syndrome, and sudden cardiac death, yet it remains a challenging diagnosis. Current guidelines recommend invasive coronary function testing to assess pathophysiology and mechanisms and to define treatment. In reality, this protocol is rarely applied, because it necessitates extended occupation of the cath lab, repetitive administration of nephrotoxic iodine contrast agents, the need for repeated testing on both coronary arteries leading to considerable radiation exposure, and significant direct expenses. The promising perspective for vasospasm testing is a noninvasive approach with advanced echocardiographic techniques, such as transthoracic Doppler echocardiography, with more sensitive indicators of ischemia. Hyperventilation and exercise tests are used for vasospasm directed testing, with assessment of the new parameters: coronary flow velocities and reserve, allowing to see deeper into macro and microvascular pathophysiology. Association between coronary flow, global longitudinal strain and microvascular dysfunction (MVD) and impaired values at hyperemia was previously demonstrated. Reduction in coronary flow velocity (CFV) despite heightened myocardial oxygen consumption and double product during hyperventilation are indicative of coronary vasospasm. Normal coronary angiography finding in patients with documented evidence of ischemia should initiate additional diagnostic testing in order to increase the yield of specific diagnosis in patients with suspected vasospasm, which could help to personalize treatment and prognosis. In order to achieve this, non-invasive provocative stress echocardiography tests should be included in the diagnostic workup. This approach, characterized by its simplicity, feasibility, safety, and efficacy, is currently undergoing extensive testing on a large scale.
DOI: https://doi.org/10.37349/ec.2024.00016

Aim:
There is a lack of studies that analyzed factors influencing on feasibility of coronary flow velocity reserve (CFVR) during exercise stress echocardiography (SE). The aim of the study was to define the feasibility of assessment of CFVR during exercise through SE depending on experience, techniques, and clinical factors.
Methods:
This is a single-center study. SE was performed using three generations of echo systems in five consecutive cohorts of patients by experienced and novice specialists. All patients performed a supine bicycle testing. CFVR was calculated in the middle/middle-distal parts of the left anterior descending artery (LAD). Three different adjustment settings were used for LAD visualization.
Results:
The study included 3,014 patients (59 years old ± 11 years old, 54% males). Age [odds ratio (OR) 0.98, 95% confidence interval (CI) 0.96–0.99, P < 0.01], body mass index (BMI; OR 0.95, 95% CI 0.91–0.98, P < 0.003), rest heart rate (OR 0.98, 95% CI 0.97–0.99, P < 0.0005) and doctor’s experience (OR 2.7, 95% CI 1.57–4.53, P < 0.0003) were independent factors that influence on feasibility. The feasibility of CFVR assessment during exercise SE in the whole population by experienced doctors was 89.4%. The feasibility of CFVR assessment of LAD in obese patients performed by experienced doctors using modern echo machines and new techniques was high (86.0%).
Conclusions:
Coronary artery velocity reserve during supine exercise SE is a feasible, non-invasive available tool. The new generation echo machine and the new techniques provide a good feasibility of CFVR assessment, even in novice doctors. Despite a lower level of possibility to assess CFVR in obese patients or with a higher resting heart rate, this method is feasible in a great majority of such patients.
Aim:
There is a lack of studies that analyzed factors influencing on feasibility of coronary flow velocity reserve (CFVR) during exercise stress echocardiography (SE). The aim of the study was to define the feasibility of assessment of CFVR during exercise through SE depending on experience, techniques, and clinical factors.
Methods:
This is a single-center study. SE was performed using three generations of echo systems in five consecutive cohorts of patients by experienced and novice specialists. All patients performed a supine bicycle testing. CFVR was calculated in the middle/middle-distal parts of the left anterior descending artery (LAD). Three different adjustment settings were used for LAD visualization.
Results:
The study included 3,014 patients (59 years old ± 11 years old, 54% males). Age [odds ratio (OR) 0.98, 95% confidence interval (CI) 0.96–0.99, P < 0.01], body mass index (BMI; OR 0.95, 95% CI 0.91–0.98, P < 0.003), rest heart rate (OR 0.98, 95% CI 0.97–0.99, P < 0.0005) and doctor’s experience (OR 2.7, 95% CI 1.57–4.53, P < 0.0003) were independent factors that influence on feasibility. The feasibility of CFVR assessment during exercise SE in the whole population by experienced doctors was 89.4%. The feasibility of CFVR assessment of LAD in obese patients performed by experienced doctors using modern echo machines and new techniques was high (86.0%).
Conclusions:
Coronary artery velocity reserve during supine exercise SE is a feasible, non-invasive available tool. The new generation echo machine and the new techniques provide a good feasibility of CFVR assessment, even in novice doctors. Despite a lower level of possibility to assess CFVR in obese patients or with a higher resting heart rate, this method is feasible in a great majority of such patients.
DOI: https://doi.org/10.37349/ec.2024.00017

Aim:
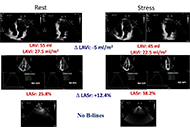
Left atrial volume index (LAVI), left atrial reservoir function through left atrial reservoir strain (LASr), and B-lines in lung ultrasound serve as supplementary indicators of left ventricular filling pressures. This study analyzes the interrelation between LAVI, LASr, and B-lines in both resting and peak vasodilator stress.
Methods:
Dipyridamole stress echocardiography (SE) was conducted on 252 individuals (180 males, 71%, age 65 years ± 10 years) with chronic coronary syndromes. LAVI was quantified using the biplane disk summation method; LASr was obtained using 2-dimensional speckle tracking echocardiography; B-lines were evaluated through a simplified 4-site scan in the third intercostal space during lung ultrasound.
Results:
During SE, a reduction in LAVI (26 ml/m2 ± 14 ml/m2 vs. 24 ml/m2 ± 12 ml/m2, P < 0.001) and an increase in LASr from rest (33% ± 8% vs. 38% ± 10%, P < 0.001) were respectively observed from rest to stress. B-lines were increased significantly during SE, from 19 (7.5%) to 29 (11.5%), P < 0.001. A substantial, inverse linear correlation was identified between LAVI and LASr both at rest (r = –0.301, P < 0.001) and peak stress (r = –0.279, P < 0.001). At group analysis, peak B-lines showed a direct correlation with peak LAVI (r = 0.151, P = 0.017) and an inverse correlation with peak LASr (r = –0.234, P < 0.001). In individual assessments, 9.7% (20/207) of patients displayed stress B-lines with normal LAVI and preserved LASr, while 20% (9/45) exhibited stress B-lines with abnormalities in both LAVI and LASr.
Conclusions:
Vasodilator SE with combined left atrial and volume assessment, related to pulmonary congestion, is feasible with a high success rate. Pulmonary congestion is more frequent with dilated left atrium with reduced atrial contractile reserve (ClinicalTrials.gov identifier: NCT030.49995; NCT050.81115).
Aim:
Left atrial volume index (LAVI), left atrial reservoir function through left atrial reservoir strain (LASr), and B-lines in lung ultrasound serve as supplementary indicators of left ventricular filling pressures. This study analyzes the interrelation between LAVI, LASr, and B-lines in both resting and peak vasodilator stress.
Methods:
Dipyridamole stress echocardiography (SE) was conducted on 252 individuals (180 males, 71%, age 65 years ± 10 years) with chronic coronary syndromes. LAVI was quantified using the biplane disk summation method; LASr was obtained using 2-dimensional speckle tracking echocardiography; B-lines were evaluated through a simplified 4-site scan in the third intercostal space during lung ultrasound.
Results:
During SE, a reduction in LAVI (26 ml/m2 ± 14 ml/m2 vs. 24 ml/m2 ± 12 ml/m2, P < 0.001) and an increase in LASr from rest (33% ± 8% vs. 38% ± 10%, P < 0.001) were respectively observed from rest to stress. B-lines were increased significantly during SE, from 19 (7.5%) to 29 (11.5%), P < 0.001. A substantial, inverse linear correlation was identified between LAVI and LASr both at rest (r = –0.301, P < 0.001) and peak stress (r = –0.279, P < 0.001). At group analysis, peak B-lines showed a direct correlation with peak LAVI (r = 0.151, P = 0.017) and an inverse correlation with peak LASr (r = –0.234, P < 0.001). In individual assessments, 9.7% (20/207) of patients displayed stress B-lines with normal LAVI and preserved LASr, while 20% (9/45) exhibited stress B-lines with abnormalities in both LAVI and LASr.
Conclusions:
Vasodilator SE with combined left atrial and volume assessment, related to pulmonary congestion, is feasible with a high success rate. Pulmonary congestion is more frequent with dilated left atrium with reduced atrial contractile reserve (ClinicalTrials.gov identifier: NCT030.49995; NCT050.81115).
DOI: https://doi.org/10.37349/ec.2024.00018

Myocardial bridging is a congenital defect characterized by the course of a segment of the coronary arteries within the heart muscle most frequently affecting the left anterior descending coronary artery (LAD). Patients with myocardial bridging may present with episodes of exertional anginal chest pain. The gold standard for non-invasive diagnosis of myocardial bridge is coronary computed tomography angiography (CCTA), which allows anatomical characterization. Coronary flow velocity reserve (CFVR) of the LAD on stress echocardiography (SE) can play an important role in the diagnosis of myocardial bridging of the LAD; a relationship between CVFR-LAD and coronary inflammation by estimating the attenuation of peri-coronary adipose tissue at CCTA has been demonstrated in patients without obstructive ischaemic heart disease. Therefore, coronary inflammation localized to the LAD has been assessed in patients diagnosed with myocardial bridging of the LAD and previous SE with CFVR-LAD in a case series.
Myocardial bridging is a congenital defect characterized by the course of a segment of the coronary arteries within the heart muscle most frequently affecting the left anterior descending coronary artery (LAD). Patients with myocardial bridging may present with episodes of exertional anginal chest pain. The gold standard for non-invasive diagnosis of myocardial bridge is coronary computed tomography angiography (CCTA), which allows anatomical characterization. Coronary flow velocity reserve (CFVR) of the LAD on stress echocardiography (SE) can play an important role in the diagnosis of myocardial bridging of the LAD; a relationship between CVFR-LAD and coronary inflammation by estimating the attenuation of peri-coronary adipose tissue at CCTA has been demonstrated in patients without obstructive ischaemic heart disease. Therefore, coronary inflammation localized to the LAD has been assessed in patients diagnosed with myocardial bridging of the LAD and previous SE with CFVR-LAD in a case series.
DOI: https://doi.org/10.37349/ec.2024.00019

Aim:
Cardio-ankle vascular index (CAVI) is a marker of arterial stiffness independent of blood pressure (BP) at the time of measurement. This work sought to evaluate the association of CAVI with left ventricular hypertrophy (LVH), a marker of long-standing hypertension (HTN) in the pediatric population.
Methods:
CAVI values from 236 children being evaluated for HTN were compared with their BP grade (normal, elevated, stage I HTN, and stage II HTN) in accordance with clinical guidelines. CAVI values were correlated to the presence of LVH and lipid profiles. One hundred seven of the studied patients had transthoracic echocardiograms available for comparison, and 126 had available lipid results. CAVI means between the groups were compared using analysis of variance.
Results:
There was no significant difference in CAVI values between the BP groups [median/interquartile range: normal BP (4.95/4.4–5.7), elevated BP (5.1/4.5–5.6), stage I/II HTN (5.0/4.3–5.5)]. Mean CAVI value was higher in the group that had LVH (5.53, standard deviation = 1.4 vs. 5.1, standard deviation = 1; P = 0.13) but was not statistically significant. However, higher mean CAVI value in children ≥ 15 years was significantly associated with the presence of LVH (5.9, standard deviation = 1.8 vs. 5.2 standard deviation = 0.8; P = 0.018).
Conclusions:
In post-pubertal children, CAVI may be a good predictor of LVH from long-standing HTN. This tool could prove useful in screening for the presence of atherosclerotic changes and provide opportunity for intervention/improved long-term outcomes.
Aim:
Cardio-ankle vascular index (CAVI) is a marker of arterial stiffness independent of blood pressure (BP) at the time of measurement. This work sought to evaluate the association of CAVI with left ventricular hypertrophy (LVH), a marker of long-standing hypertension (HTN) in the pediatric population.
Methods:
CAVI values from 236 children being evaluated for HTN were compared with their BP grade (normal, elevated, stage I HTN, and stage II HTN) in accordance with clinical guidelines. CAVI values were correlated to the presence of LVH and lipid profiles. One hundred seven of the studied patients had transthoracic echocardiograms available for comparison, and 126 had available lipid results. CAVI means between the groups were compared using analysis of variance.
Results:
There was no significant difference in CAVI values between the BP groups [median/interquartile range: normal BP (4.95/4.4–5.7), elevated BP (5.1/4.5–5.6), stage I/II HTN (5.0/4.3–5.5)]. Mean CAVI value was higher in the group that had LVH (5.53, standard deviation = 1.4 vs. 5.1, standard deviation = 1; P = 0.13) but was not statistically significant. However, higher mean CAVI value in children ≥ 15 years was significantly associated with the presence of LVH (5.9, standard deviation = 1.8 vs. 5.2 standard deviation = 0.8; P = 0.018).
Conclusions:
In post-pubertal children, CAVI may be a good predictor of LVH from long-standing HTN. This tool could prove useful in screening for the presence of atherosclerotic changes and provide opportunity for intervention/improved long-term outcomes.
DOI: https://doi.org/10.37349/ec.2024.00020
This article belongs to the special issue Environmental Cardiology

Aim:
The DNA of the atheroma is hypermethylated relative to adjacent healthy vascular tissue. A significant portion of hypermethylated loci in the atheroma DNA map to genes related to macrophage function. Reversing macrophage DNA methylation to physiological levels by targeting DNA methyltransferase (DNMT) activity may therefore slow atherogenesis. Here, the anti-inflammatory and anti-atherogenic activity of macrophage-targeted DNMT inhibitor SGI-1027 were tested.
Methods:
SGI-1027 was encapsulated into human serum albumin (HSA) nanoparticle (HSANP) functionalized with the PP1 peptide, a macrophage scavenger receptor 1 ligand, fused to a FLAG epitope (S-HSANP-FLAGPP1).
Results:
Nanoparticle physico-chemical characteristics predicted good marginalization towards the vascular wall, although SGI-1027 encapsulation efficiency was relatively low (~23%). S-HSANP-FLAGPP1 were rapidly internalized compared to non-functionalized and, surprisingly, functionalized void controls, and induced a shift towards an anti-inflammatory profile of secreted cytokines in human THP-1 macrophages. S-HSANP-FLAGPP1 colonized the atheroma and induced a significant ~44% reduction of atherosclerosis burden in the aortic tree of apolipoprotein E (ApoE)-null mice compared to controls. A reduction in aortic root atherosclerosis was observed, although primarily induced by HSANP irrespective of loading or functionalization. No alteration of body weight, non-vascular tissue gross histology, plasma glucose, triglyceride or cholesterol were observed. HSA whether free or structured in nanoparticles, induced a 3–4-fold increase in high-density lipoprotein (HDL) compared to vehicle.
Conclusions:
Unexpectedly, effects that were likely non-epigenetic and induced by HSA per se were observed. HSANP loaded with SGI-1027 were anti-atherogenic but in an anatomical location-dependent fashion. SGI-1027 displayed a novel anti-inflammatory activity in non-proliferating THP-1 cells, implying that those effects are likely unrelated to DNMT inhibition. HSA elevated HDL per se, thus underlining a possible additional advantage of HSA-based nanocarriers.
Aim:
The DNA of the atheroma is hypermethylated relative to adjacent healthy vascular tissue. A significant portion of hypermethylated loci in the atheroma DNA map to genes related to macrophage function. Reversing macrophage DNA methylation to physiological levels by targeting DNA methyltransferase (DNMT) activity may therefore slow atherogenesis. Here, the anti-inflammatory and anti-atherogenic activity of macrophage-targeted DNMT inhibitor SGI-1027 were tested.
Methods:
SGI-1027 was encapsulated into human serum albumin (HSA) nanoparticle (HSANP) functionalized with the PP1 peptide, a macrophage scavenger receptor 1 ligand, fused to a FLAG epitope (S-HSANP-FLAGPP1).
Results:
Nanoparticle physico-chemical characteristics predicted good marginalization towards the vascular wall, although SGI-1027 encapsulation efficiency was relatively low (~23%). S-HSANP-FLAGPP1 were rapidly internalized compared to non-functionalized and, surprisingly, functionalized void controls, and induced a shift towards an anti-inflammatory profile of secreted cytokines in human THP-1 macrophages. S-HSANP-FLAGPP1 colonized the atheroma and induced a significant ~44% reduction of atherosclerosis burden in the aortic tree of apolipoprotein E (ApoE)-null mice compared to controls. A reduction in aortic root atherosclerosis was observed, although primarily induced by HSANP irrespective of loading or functionalization. No alteration of body weight, non-vascular tissue gross histology, plasma glucose, triglyceride or cholesterol were observed. HSA whether free or structured in nanoparticles, induced a 3–4-fold increase in high-density lipoprotein (HDL) compared to vehicle.
Conclusions:
Unexpectedly, effects that were likely non-epigenetic and induced by HSA per se were observed. HSANP loaded with SGI-1027 were anti-atherogenic but in an anatomical location-dependent fashion. SGI-1027 displayed a novel anti-inflammatory activity in non-proliferating THP-1 cells, implying that those effects are likely unrelated to DNMT inhibition. HSA elevated HDL per se, thus underlining a possible additional advantage of HSA-based nanocarriers.
DOI: https://doi.org/10.37349/ec.2024.00021

Aim:
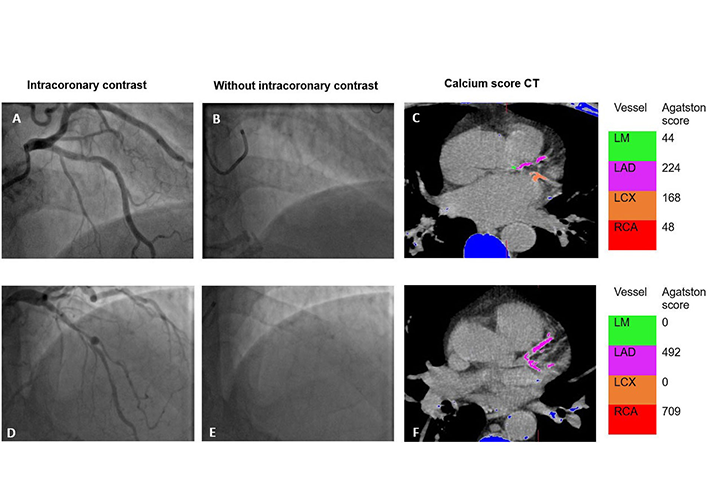
The pattern and severity of coronary artery calcification (CAC) can influence prognosis and outcome in percutaneous coronary intervention. An objective assessment of CAC during invasive angiography may provide additional prognostic information. This study aimed to assess the correlation between the angiographic Birmingham calcium score (BCS) and the Agatston coronary calcium score (CCS) performed as part of single-photon emission computed tomography myocardial perfusion imaging (SPECT-MPI).
Methods:
In this retrospective observational study, patients undergoing SPECT-MPI and invasive coronary angiography as part of their routine management were included. BCS was calculated by reviewing angiography images in retrospect by an observer blinded to the SPECT-MPI calcium score. Spearman’s correlation was used to analyze the correlation between BCS and SPECT-MPI. Receiver operating characteristic curve was used to detect cut-off for BCS that would detect clinically significant CAC [> 400 Agatston units (AU)]. Kaplan-Meier was used to report on outcomes at 5 years follow-up.
Results:
In this cohort of 151 patients, there was a positive correlation between BCS and CCS [Spearman correlation coefficient (r) = 0.558, P < 0.001]. Cumulative BCS of 1 was able to identify clinically significant CAC [area under the curve 0.788, 95% confidence interval (CI) 0.714–0.863]. Cumulative BCS ≥ 3 was associated with major adverse outcomes at 5 years follow-up (log rank P = 0.013).
Conclusion:
BCS correlates well with established higher CCS. Application of BCS during invasive coronary angiography will aid risk stratification, management, and follow-up with no extra patient involvement, radiation, or costs.
Aim:
The pattern and severity of coronary artery calcification (CAC) can influence prognosis and outcome in percutaneous coronary intervention. An objective assessment of CAC during invasive angiography may provide additional prognostic information. This study aimed to assess the correlation between the angiographic Birmingham calcium score (BCS) and the Agatston coronary calcium score (CCS) performed as part of single-photon emission computed tomography myocardial perfusion imaging (SPECT-MPI).
Methods:
In this retrospective observational study, patients undergoing SPECT-MPI and invasive coronary angiography as part of their routine management were included. BCS was calculated by reviewing angiography images in retrospect by an observer blinded to the SPECT-MPI calcium score. Spearman’s correlation was used to analyze the correlation between BCS and SPECT-MPI. Receiver operating characteristic curve was used to detect cut-off for BCS that would detect clinically significant CAC [> 400 Agatston units (AU)]. Kaplan-Meier was used to report on outcomes at 5 years follow-up.
Results:
In this cohort of 151 patients, there was a positive correlation between BCS and CCS [Spearman correlation coefficient (r) = 0.558, P < 0.001]. Cumulative BCS of 1 was able to identify clinically significant CAC [area under the curve 0.788, 95% confidence interval (CI) 0.714–0.863]. Cumulative BCS ≥ 3 was associated with major adverse outcomes at 5 years follow-up (log rank P = 0.013).
Conclusion:
BCS correlates well with established higher CCS. Application of BCS during invasive coronary angiography will aid risk stratification, management, and follow-up with no extra patient involvement, radiation, or costs.
DOI: https://doi.org/10.37349/ec.2024.00022

Left ventricular (LV) function is typically evaluated through LV ejection fraction (EF), a robust indicator of risk, showing a nonlinear increase in mortality rates below 40%. Conversely, excessively high EF values (> 65%) also correlate with elevated mortality, following a U-shaped curve, with its nadir observed between 50% and 65%. This underscores the necessity for improved identification of the hypercontractile phenotype. However, EF is not synonymous with LV contraction function, as it can fluctuate independently of contractility due to variations in afterload, preload, heart rate, and ventricular-arterial coupling. Assessing the contractile status of the LV requires more specific metrics, such as LV elastance (or contractile force) and global longitudinal strain. Current guidelines outline various parameters for a more precise characterization of LV contractility, yet further research is warranted for validation. The true hypercontractile phenotype is evident in cardiac pathologies such as hypertrophic cardiomyopathy, ischemia with angiographically normal coronary arteries, Tako-tsubo syndrome, heart failure with preserved EF, and may also stem from systemic disorders including anemia, hyperthyroidism, liver, kidney, or pulmonary diseases. The hypercontractile phenotype constitutes a distinctive hemodynamic substrate underlying clinical manifestations such as angina, dyspnea, or arrhythmias, presenting a target for intervention through beta-blockers or specific cardiac myosin inhibitors. While EF remains pivotal for clinical classification, risk stratification, and therapeutic decision-making, integrating it with other indices of LV function can enhance the characterization of the hypercontractile phenotype.
Left ventricular (LV) function is typically evaluated through LV ejection fraction (EF), a robust indicator of risk, showing a nonlinear increase in mortality rates below 40%. Conversely, excessively high EF values (> 65%) also correlate with elevated mortality, following a U-shaped curve, with its nadir observed between 50% and 65%. This underscores the necessity for improved identification of the hypercontractile phenotype. However, EF is not synonymous with LV contraction function, as it can fluctuate independently of contractility due to variations in afterload, preload, heart rate, and ventricular-arterial coupling. Assessing the contractile status of the LV requires more specific metrics, such as LV elastance (or contractile force) and global longitudinal strain. Current guidelines outline various parameters for a more precise characterization of LV contractility, yet further research is warranted for validation. The true hypercontractile phenotype is evident in cardiac pathologies such as hypertrophic cardiomyopathy, ischemia with angiographically normal coronary arteries, Tako-tsubo syndrome, heart failure with preserved EF, and may also stem from systemic disorders including anemia, hyperthyroidism, liver, kidney, or pulmonary diseases. The hypercontractile phenotype constitutes a distinctive hemodynamic substrate underlying clinical manifestations such as angina, dyspnea, or arrhythmias, presenting a target for intervention through beta-blockers or specific cardiac myosin inhibitors. While EF remains pivotal for clinical classification, risk stratification, and therapeutic decision-making, integrating it with other indices of LV function can enhance the characterization of the hypercontractile phenotype.
DOI: https://doi.org/10.37349/ec.2024.00025

Metabolic syndrome (MetS) is known as a non-communicable disease (NCD) that affects more and more individuals. MetS is closely related to type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD), obesity and inflammation. It is associated with T2DM due to the disturbance in insulin secretion/effect, eventually leading to insulin resistance (IR). The link between MetS and CVD is due to accelerated atherosclerosis in response to chronic inflammation. This literature review was based on a search in the PubMed database. All selected articles are written in English and cover a period of approximately 10 years (January 2014 to May 2023). The first selection used MeSH terms such as: “metabolic syndrome”, “type 2 diabetes mellitus”, “obesity”, “inflammation”, and “insulin resistance” and different associations between them. Titles and abstracts were analyzed. In the end, 44 articles were selected, 4 of which were meta-analysis studies. Currently, an individual is considered to have MetS if they present 3 of the following changes: increased waist circumference, increased triglycerides (TG), reduced high-density lipoprotein cholesterol (HDL-C), increased fasting blood glucose and hypertension. We believe this can often lead to a false diagnosis. The objective of this paper is to compile what we consider to be an appropriate panel of MetS indicators. The markers that stand out in this review are the lipid profile, anti- and pro-inflammatory function and oxidative stress. Considering the research, we believe that a complete panel, to correlate the most characteristic conditions of MetS, should include the following markers: TG/HDL-C ratio, small dense low-density lipoprotein cholesterol (SdLDL-C), lipid peroxidation markers, leptin/adiponectin ratio, plasminogen activator inhibitor-1 (PAI-1), activin-A and ferritin levels. Finally, it is important to expand research on the pathophysiology of MetS and confirm the most appropriate markers as well as discover new ones to correctly diagnose this condition.
Metabolic syndrome (MetS) is known as a non-communicable disease (NCD) that affects more and more individuals. MetS is closely related to type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD), obesity and inflammation. It is associated with T2DM due to the disturbance in insulin secretion/effect, eventually leading to insulin resistance (IR). The link between MetS and CVD is due to accelerated atherosclerosis in response to chronic inflammation. This literature review was based on a search in the PubMed database. All selected articles are written in English and cover a period of approximately 10 years (January 2014 to May 2023). The first selection used MeSH terms such as: “metabolic syndrome”, “type 2 diabetes mellitus”, “obesity”, “inflammation”, and “insulin resistance” and different associations between them. Titles and abstracts were analyzed. In the end, 44 articles were selected, 4 of which were meta-analysis studies. Currently, an individual is considered to have MetS if they present 3 of the following changes: increased waist circumference, increased triglycerides (TG), reduced high-density lipoprotein cholesterol (HDL-C), increased fasting blood glucose and hypertension. We believe this can often lead to a false diagnosis. The objective of this paper is to compile what we consider to be an appropriate panel of MetS indicators. The markers that stand out in this review are the lipid profile, anti- and pro-inflammatory function and oxidative stress. Considering the research, we believe that a complete panel, to correlate the most characteristic conditions of MetS, should include the following markers: TG/HDL-C ratio, small dense low-density lipoprotein cholesterol (SdLDL-C), lipid peroxidation markers, leptin/adiponectin ratio, plasminogen activator inhibitor-1 (PAI-1), activin-A and ferritin levels. Finally, it is important to expand research on the pathophysiology of MetS and confirm the most appropriate markers as well as discover new ones to correctly diagnose this condition.
DOI: https://doi.org/10.37349/ec.2024.00026
This article belongs to the special issue Molecular Mechanisms of Cardiovascular Aging

Adventitial crosslinking is a method in current investigational stage for preventing the rupture of aortic aneurysms. It is based on the photochemical crosslinking of adventitial collagen by exposure to ultraviolet A radiation. Essentially, an adventitial top layer is generated that displays enhanced mechanical properties and imparts additional strength and stiffness to the aneurysmal wall. Looking back upon the history of aortic surgery during 1940s, the aortic film wrapping, then dubbed “cellophane wrapping”, also was a procedure employed for delaying the aneurysmal rupture. In principle, the two procedures are similar in that both result in laminar composites, although the top layers differ fundamentally from each other. This review discussed in some detail the use and clinical outcomes of the aortic wrapping with artificial films, also mentioning the contemporary procedures still grouped under this umbrella term. The focus of the review was a comparative view on two procedures, the aortic film wrapping and adventitial crosslinking. It was concluded that the methods are different in many aspects, including the mechanisms of action. In fact, the promoters of adventitial crosslinking were not aware of the prior existence of aortic film wrapping. However, the achievements of the classical wrapping, by now regarded as merely historical episodes, did not discard prior knowledge, but repurposed it in a process that led to innovative strategies.
Adventitial crosslinking is a method in current investigational stage for preventing the rupture of aortic aneurysms. It is based on the photochemical crosslinking of adventitial collagen by exposure to ultraviolet A radiation. Essentially, an adventitial top layer is generated that displays enhanced mechanical properties and imparts additional strength and stiffness to the aneurysmal wall. Looking back upon the history of aortic surgery during 1940s, the aortic film wrapping, then dubbed “cellophane wrapping”, also was a procedure employed for delaying the aneurysmal rupture. In principle, the two procedures are similar in that both result in laminar composites, although the top layers differ fundamentally from each other. This review discussed in some detail the use and clinical outcomes of the aortic wrapping with artificial films, also mentioning the contemporary procedures still grouped under this umbrella term. The focus of the review was a comparative view on two procedures, the aortic film wrapping and adventitial crosslinking. It was concluded that the methods are different in many aspects, including the mechanisms of action. In fact, the promoters of adventitial crosslinking were not aware of the prior existence of aortic film wrapping. However, the achievements of the classical wrapping, by now regarded as merely historical episodes, did not discard prior knowledge, but repurposed it in a process that led to innovative strategies.
DOI: https://doi.org/10.37349/ec.2024.00027

Pulmonary embolism (PE) is the third leading cause of cardiovascular mortality worldwide. Percutaneous mechanical thrombectomy is indicated in patients with contraindications to thrombolysis. Complications include atrial or ventricular perforation causing tamponade. We describe one case of pericardial tamponade in an elderly woman who underwent thrombectomy for acute PE. An 88-year-old woman presented with acute shortness of breath. She was tachycardic, oxygen saturation of 80% and blood pressure of 95/57 mmHg. Bedside ultrasound showed a dilated right ventricle. Electrocardiogram showed large S wave in lead I, Q wave and inverted T wave in lead III. Computed tomography (CT) angiogram of the chest revealed an extensive saddle PE. Tissue plasminogen activator was deferred given patient’s age. Full dose anticoagulation was started and she underwent a successful percutaneous thrombectomy with FlowTriever device. Two hours later, she developed severe back pain and hypotension to 88/63 mmHg. Hemoglobin dropped from 13.7 g/dL to 8.8 g/dL. CT chest angiogram showed dense pericardial effusion, likely hemopericardium, with mass effect on the heart. Bedside pericardiocentesis was attempted and converted to pericardial window given sustained hypotension. She went into cardiac arrest. Emergent thoracotomy revealed significant hemothorax. The pericardium was opened yielding a blue, globally ischemic, and non-contracting heart. Cardiac massage and intra-cardiac epinephrine attempted unsuccessfully. Percutaneous thrombectomy using the large bore design FlowTriever system has shown promising results in reducing clot burden and improving hemodynamics. Attention must be paid to life threatening complications such as cardiac tamponade which can be precipitated by using these devices.
Pulmonary embolism (PE) is the third leading cause of cardiovascular mortality worldwide. Percutaneous mechanical thrombectomy is indicated in patients with contraindications to thrombolysis. Complications include atrial or ventricular perforation causing tamponade. We describe one case of pericardial tamponade in an elderly woman who underwent thrombectomy for acute PE. An 88-year-old woman presented with acute shortness of breath. She was tachycardic, oxygen saturation of 80% and blood pressure of 95/57 mmHg. Bedside ultrasound showed a dilated right ventricle. Electrocardiogram showed large S wave in lead I, Q wave and inverted T wave in lead III. Computed tomography (CT) angiogram of the chest revealed an extensive saddle PE. Tissue plasminogen activator was deferred given patient’s age. Full dose anticoagulation was started and she underwent a successful percutaneous thrombectomy with FlowTriever device. Two hours later, she developed severe back pain and hypotension to 88/63 mmHg. Hemoglobin dropped from 13.7 g/dL to 8.8 g/dL. CT chest angiogram showed dense pericardial effusion, likely hemopericardium, with mass effect on the heart. Bedside pericardiocentesis was attempted and converted to pericardial window given sustained hypotension. She went into cardiac arrest. Emergent thoracotomy revealed significant hemothorax. The pericardium was opened yielding a blue, globally ischemic, and non-contracting heart. Cardiac massage and intra-cardiac epinephrine attempted unsuccessfully. Percutaneous thrombectomy using the large bore design FlowTriever system has shown promising results in reducing clot burden and improving hemodynamics. Attention must be paid to life threatening complications such as cardiac tamponade which can be precipitated by using these devices.
DOI: https://doi.org/10.37349/ec.2024.00028

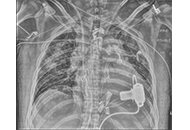
Aim:
Temporary right ventricular assist device (t-RVAD) is an option for those patients in right ventricular failure (RVF) after left ventricular assist device (LVAD) resistant to ionotropic therapy. There are two options to placing a t-RVAD: an open, central technique or a percutaneous placement with Protek Duo® cannula.
Methods:
We compare these two t-RVAD devices that treat RVF after LVAD placement. Between 2013–2019, 22 patients were identified needing t-RVAD support after LVAD placement. Fourteen patients had open/central while 8 patients had percutaneous right ventricular assist device (RVAD) support.
Results:
There was no difference in length of ICU stay (49 ± 32 days Protek Duo® vs. 45 ± 22 days “open”; P = 0.73); hospital length of stay (57 ± 39 days vs. 55 ± 28 days; P = 0.088); discharge from ICU and hospital (62.1% Protek Duo® vs. 57% for “open”; P = 0.9 for both); or the one-year survival between the two groups (62% Protek Duo® vs. 50% “open”; P = 0.67). The Protek Duo® group had less total time on the ventilator (15 ± 9 days vs. 27 ± 17 days; P = 0.044) and required less amount of blood products (17 ± 8.9 units RBC and 2.0 ± 1.91 units FFP vs. 31 ± 20.5 units RBC and 11.5 ± 10 units FFP; P = 0.046 and P = 0.005).
Conclusions:
Percutaneous t-RVAD support is a viable option for patients whom undergo LVAD placement and require right ventricular mechanical support.
Aim:
Temporary right ventricular assist device (t-RVAD) is an option for those patients in right ventricular failure (RVF) after left ventricular assist device (LVAD) resistant to ionotropic therapy. There are two options to placing a t-RVAD: an open, central technique or a percutaneous placement with Protek Duo® cannula.
Methods:
We compare these two t-RVAD devices that treat RVF after LVAD placement. Between 2013–2019, 22 patients were identified needing t-RVAD support after LVAD placement. Fourteen patients had open/central while 8 patients had percutaneous right ventricular assist device (RVAD) support.
Results:
There was no difference in length of ICU stay (49 ± 32 days Protek Duo® vs. 45 ± 22 days “open”; P = 0.73); hospital length of stay (57 ± 39 days vs. 55 ± 28 days; P = 0.088); discharge from ICU and hospital (62.1% Protek Duo® vs. 57% for “open”; P = 0.9 for both); or the one-year survival between the two groups (62% Protek Duo® vs. 50% “open”; P = 0.67). The Protek Duo® group had less total time on the ventilator (15 ± 9 days vs. 27 ± 17 days; P = 0.044) and required less amount of blood products (17 ± 8.9 units RBC and 2.0 ± 1.91 units FFP vs. 31 ± 20.5 units RBC and 11.5 ± 10 units FFP; P = 0.046 and P = 0.005).
Conclusions:
Percutaneous t-RVAD support is a viable option for patients whom undergo LVAD placement and require right ventricular mechanical support.
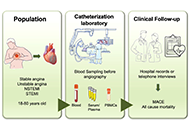
DOI: https://doi.org/10.37349/ec.2024.00029

Vascular aging is recognized as one of the hallmarks of atherosclerosis. Currently, a growing body of evidence suggests that there exists a mutual crosstalk between telomere dysfunction and mitochondrial dysmetabolism during the process of vascular senescence. This underscores the importance of comprehensively studying the molecular mediators involved in this complex and intricate connection. In pursuit of this goal, the “VICTORIA” protocol entails a prospective single-center cohort study aimed at recruiting patients undergoing coronary angiography at Niguarda Hospital in Italy. The primary objective is to explore potential associations between peripheral markers of cell aging (telomere length and mtDNA content), dysregulation of non-coding RNA [specifically lncRNA TERRA and mitochondrial microRNA (MitomiR)], and the varied presentations of ischemic heart disease (stable angina, unstable angina, NSTEMI, and STEMI). Furthermore, we aim to investigate whether these markers correlate with vulnerable plaque characteristics, as assessed by optical coherence tomography findings. Additionally, systemic levels of pro-inflammatory biomarkers and novel indicators of senescence will be assessed. Patients will be followed up at 1 year to monitor primary outcomes including mortality, myocardial infarction, stroke, unplanned revascularization, and rehospitalization. The anticipated findings of this study hold promise for advancing our understanding of the telomere-mitochondria crosstalk, potentially paving the way for novel treatment modalities and refined risk stratification approaches for acute coronary syndrome.
Vascular aging is recognized as one of the hallmarks of atherosclerosis. Currently, a growing body of evidence suggests that there exists a mutual crosstalk between telomere dysfunction and mitochondrial dysmetabolism during the process of vascular senescence. This underscores the importance of comprehensively studying the molecular mediators involved in this complex and intricate connection. In pursuit of this goal, the “VICTORIA” protocol entails a prospective single-center cohort study aimed at recruiting patients undergoing coronary angiography at Niguarda Hospital in Italy. The primary objective is to explore potential associations between peripheral markers of cell aging (telomere length and mtDNA content), dysregulation of non-coding RNA [specifically lncRNA TERRA and mitochondrial microRNA (MitomiR)], and the varied presentations of ischemic heart disease (stable angina, unstable angina, NSTEMI, and STEMI). Furthermore, we aim to investigate whether these markers correlate with vulnerable plaque characteristics, as assessed by optical coherence tomography findings. Additionally, systemic levels of pro-inflammatory biomarkers and novel indicators of senescence will be assessed. Patients will be followed up at 1 year to monitor primary outcomes including mortality, myocardial infarction, stroke, unplanned revascularization, and rehospitalization. The anticipated findings of this study hold promise for advancing our understanding of the telomere-mitochondria crosstalk, potentially paving the way for novel treatment modalities and refined risk stratification approaches for acute coronary syndrome.
DOI: https://doi.org/10.37349/ec.2024.00030
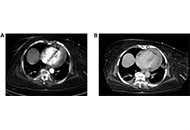
This article belongs to the special issue Molecular Mechanisms of Cardiovascular Aging

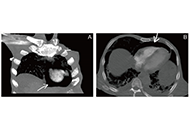
Pneumopericardium, the presence of air within the pericardial sac, is a rare but critical condition that can lead to severe complications such as tension pneumopericardium and cardiac tamponade, causing hemodynamic instability and necessitating immediate intervention. Various etiologies include congenital defects, post-surgical complications, infections, and trauma. Malignancies, such as advanced esophageal cancer or lung carcinoma, can also cause pneumopericardium via fistula formation. Multimodal imaging, including chest X-ray, echocardiography, and computed tomography (CT), is essential for diagnosis. This case report discusses a 65-year-old male with advanced pancreatic adenocarcinoma who developed pneumopericardium following the removal of a left lobe liver drainage catheter. Initial CT imaging revealed liver lesions suspicious for metastatic disease or abscess, leading to drainage procedures. Following the removal of the drainage tube, the patient experienced respiratory distress and hypotension, and computed tomography pulmonary angiogram (CTPA) revealed pneumopericardium, likely due to a fistula formed between the abscess and pericardium. Despite no echocardiographic signs of tamponade, the patient’s persistent hypotension warranted CT-guided pericardiocentesis, resulting in gradual blood pressure improvement. This case highlights the intricate interplay between malignancy, infection, and procedural complications in developing pneumopericardium. It emphasizes the need for a multidisciplinary approach and the importance of considering both the quantity and rate of air accumulation when assessing the risk of hemodynamic compromise. The patient’s hemodynamic instability and subsequent improvement following pericardiocentesis underscore the critical role of timely intervention in managing this condition.
Pneumopericardium, the presence of air within the pericardial sac, is a rare but critical condition that can lead to severe complications such as tension pneumopericardium and cardiac tamponade, causing hemodynamic instability and necessitating immediate intervention. Various etiologies include congenital defects, post-surgical complications, infections, and trauma. Malignancies, such as advanced esophageal cancer or lung carcinoma, can also cause pneumopericardium via fistula formation. Multimodal imaging, including chest X-ray, echocardiography, and computed tomography (CT), is essential for diagnosis. This case report discusses a 65-year-old male with advanced pancreatic adenocarcinoma who developed pneumopericardium following the removal of a left lobe liver drainage catheter. Initial CT imaging revealed liver lesions suspicious for metastatic disease or abscess, leading to drainage procedures. Following the removal of the drainage tube, the patient experienced respiratory distress and hypotension, and computed tomography pulmonary angiogram (CTPA) revealed pneumopericardium, likely due to a fistula formed between the abscess and pericardium. Despite no echocardiographic signs of tamponade, the patient’s persistent hypotension warranted CT-guided pericardiocentesis, resulting in gradual blood pressure improvement. This case highlights the intricate interplay between malignancy, infection, and procedural complications in developing pneumopericardium. It emphasizes the need for a multidisciplinary approach and the importance of considering both the quantity and rate of air accumulation when assessing the risk of hemodynamic compromise. The patient’s hemodynamic instability and subsequent improvement following pericardiocentesis underscore the critical role of timely intervention in managing this condition.
DOI: https://doi.org/10.37349/ec.2024.00031

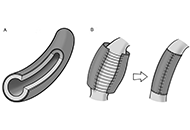
Redo surgical aortic valve replacement (SAVR) has long been the standard treatment for severe symptomatic aortic stenosis (AS) and insufficiency, but transcatheter aortic valve replacement (TAVR) has emerged as a less invasive alternative, particularly for symptomatic AS. With calcification being a primary mechanism behind structural valve degeneration, factors such as patient characteristics and the type of bioprosthetic valve play crucial roles in determining risk. Valve-in-valve TAVR (ViV TAVR) has gained prominence as a viable alternative to redo SAVR in recent years. Echocardiography plays a pivotal role in patient selection, procedural guidance, and post-procedural evaluation in ViV TAVR. From pre-procedural assessment using transthoracic and transesophageal echocardiography to intra-procedural guidance and post-procedural follow-up, echocardiography aids in ensuring procedural success and monitoring valve function. Decision-making between redo SAVR and ViV TAVR involves multidisciplinary teams considering various factors like patient risk profile, anatomical considerations, and technical feasibility. While redo SAVR remains preferred for younger patients with lower risk, ViV TAVR is increasingly considered for older patients or those with higher reoperation risks. Factors such as coronary artery obstruction risk, patient-prosthesis mismatch, and paravalvular leak guide treatment decisions. Procedural techniques in ViV TAVR have evolved to minimize complications and optimize outcomes. Factors like access route selection, valve choice, and procedural guidance techniques significantly impact procedural success. Balloon valve fracture and preventive strategies against coronary obstruction are crucial considerations during the procedure. Post-procedural evaluation involves assessing clinical and hemodynamic outcomes, with long-term studies indicating favorable results but highlighting the importance of careful evaluation of transprosthetic gradients and valve function. ViV TAVR offers promising outcomes but requires meticulous patient selection, procedural planning, and post-procedural management to ensure optimal results.
Redo surgical aortic valve replacement (SAVR) has long been the standard treatment for severe symptomatic aortic stenosis (AS) and insufficiency, but transcatheter aortic valve replacement (TAVR) has emerged as a less invasive alternative, particularly for symptomatic AS. With calcification being a primary mechanism behind structural valve degeneration, factors such as patient characteristics and the type of bioprosthetic valve play crucial roles in determining risk. Valve-in-valve TAVR (ViV TAVR) has gained prominence as a viable alternative to redo SAVR in recent years. Echocardiography plays a pivotal role in patient selection, procedural guidance, and post-procedural evaluation in ViV TAVR. From pre-procedural assessment using transthoracic and transesophageal echocardiography to intra-procedural guidance and post-procedural follow-up, echocardiography aids in ensuring procedural success and monitoring valve function. Decision-making between redo SAVR and ViV TAVR involves multidisciplinary teams considering various factors like patient risk profile, anatomical considerations, and technical feasibility. While redo SAVR remains preferred for younger patients with lower risk, ViV TAVR is increasingly considered for older patients or those with higher reoperation risks. Factors such as coronary artery obstruction risk, patient-prosthesis mismatch, and paravalvular leak guide treatment decisions. Procedural techniques in ViV TAVR have evolved to minimize complications and optimize outcomes. Factors like access route selection, valve choice, and procedural guidance techniques significantly impact procedural success. Balloon valve fracture and preventive strategies against coronary obstruction are crucial considerations during the procedure. Post-procedural evaluation involves assessing clinical and hemodynamic outcomes, with long-term studies indicating favorable results but highlighting the importance of careful evaluation of transprosthetic gradients and valve function. ViV TAVR offers promising outcomes but requires meticulous patient selection, procedural planning, and post-procedural management to ensure optimal results.
DOI: https://doi.org/10.37349/ec.2024.00032

The clinical manifestations of COVID-19 which mainly involve the respiratory system may however affect also cardiovascular system. There are a lot and still increasing numbers of reports revealing cardiovascular complications of COVID-19, which may occur in the acute phase as well as during longer follow-up period. The most clinically important diseases include: pulmonary embolism (PE), myocarditis, and acute coronary syndromes (ACS) as well as arrhythmias with the very common atrial fibrillation (AF) and pericarditis. In this review, cardiac imaging options in patients with and after coronavirus infection are presented, showing potential utility for expanding and improving the full and accurate diagnosis of potential complications. Echocardiography, magnetic resonance imaging, and computed tomography (CT) are considered in turn, highlighting their best advantages in patients affected by COVID.
The clinical manifestations of COVID-19 which mainly involve the respiratory system may however affect also cardiovascular system. There are a lot and still increasing numbers of reports revealing cardiovascular complications of COVID-19, which may occur in the acute phase as well as during longer follow-up period. The most clinically important diseases include: pulmonary embolism (PE), myocarditis, and acute coronary syndromes (ACS) as well as arrhythmias with the very common atrial fibrillation (AF) and pericarditis. In this review, cardiac imaging options in patients with and after coronavirus infection are presented, showing potential utility for expanding and improving the full and accurate diagnosis of potential complications. Echocardiography, magnetic resonance imaging, and computed tomography (CT) are considered in turn, highlighting their best advantages in patients affected by COVID.
DOI: https://doi.org/10.37349/ec.2023.00008

Mitral valve prolapse (MVP) is a relatively common mitral valvulopathy and the most common cause of isolated primary mitral regurgitation (MR) requiring surgical repair. It affects about 1–3% of the general population. Although MVP is viewed as a benign condition, the association between MVP and sudden cardiac death (SCD) has been proven. Patients with MVP have a three times higher risk of SCD than the general population. The underlying mechanisms and predictors of arrhythmias, which occur in patients with MVP, are still poorly understood. However, some echocardiographic features such as mitral annulus disjunction (MAD), bileaflet MVP (biMVP), and papillary muscle (PM) fibrosis were frequently linked with increased number of arrhythmic events and are referred to as “arrhythmogenic” or “malignant”. Arrhythmogenic MVP (AMVP) has also been associated with other factors such as female sex, polymorphic premature ventricular contraction (PVC), abnormalities of T-waves, and Pickelhaube sign on tissue Doppler tracing of the lateral part of the mitral annulus. Cardiac magnetic resonance (CMR) imaging and speckle tracking echocardiography are new tools showing significant potential for detection of malignant features of AMVP. This paper presents various data coming from electrocardiography (ECG) analysis, echocardiography, and other imaging techniques as well as compilation of the recent studies on the subject of MVP.
Mitral valve prolapse (MVP) is a relatively common mitral valvulopathy and the most common cause of isolated primary mitral regurgitation (MR) requiring surgical repair. It affects about 1–3% of the general population. Although MVP is viewed as a benign condition, the association between MVP and sudden cardiac death (SCD) has been proven. Patients with MVP have a three times higher risk of SCD than the general population. The underlying mechanisms and predictors of arrhythmias, which occur in patients with MVP, are still poorly understood. However, some echocardiographic features such as mitral annulus disjunction (MAD), bileaflet MVP (biMVP), and papillary muscle (PM) fibrosis were frequently linked with increased number of arrhythmic events and are referred to as “arrhythmogenic” or “malignant”. Arrhythmogenic MVP (AMVP) has also been associated with other factors such as female sex, polymorphic premature ventricular contraction (PVC), abnormalities of T-waves, and Pickelhaube sign on tissue Doppler tracing of the lateral part of the mitral annulus. Cardiac magnetic resonance (CMR) imaging and speckle tracking echocardiography are new tools showing significant potential for detection of malignant features of AMVP. This paper presents various data coming from electrocardiography (ECG) analysis, echocardiography, and other imaging techniques as well as compilation of the recent studies on the subject of MVP.
DOI: https://doi.org/10.37349/ec.2023.00009
This article belongs to the special issue Common cardiovascular target for a wide gamut of contemporary health problems – thrombotic and arrhythmic sides of an inflammatory coin

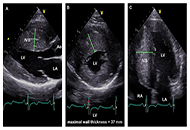
The use of echocardiography, a straightforward and widely available technique, allows for a comprehensive assessment of the patient with hypertrophic cardiomyopathy (HCM) under both resting and stress conditions. The true prevalence of HCM has been redefined over time by this imaging approach, which has also made it feasible to pinpoint parameters that clinicians may use to stratify patients at risk for adverse cardiovascular events. The current and emerging prognostic predictors in HCM, assessed with transthoracic echocardiography at rest and during provocation, are discussed in this review.
The use of echocardiography, a straightforward and widely available technique, allows for a comprehensive assessment of the patient with hypertrophic cardiomyopathy (HCM) under both resting and stress conditions. The true prevalence of HCM has been redefined over time by this imaging approach, which has also made it feasible to pinpoint parameters that clinicians may use to stratify patients at risk for adverse cardiovascular events. The current and emerging prognostic predictors in HCM, assessed with transthoracic echocardiography at rest and during provocation, are discussed in this review.
DOI: https://doi.org/10.37349/ec.2023.00010
This article belongs to the special issue Common cardiovascular target for a wide gamut of contemporary health problems – thrombotic and arrhythmic sides of an inflammatory coin

