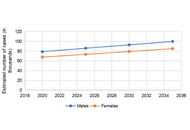












According to the Global Cancer Observatory (GLOBOCAN) 2020, colorectal carcinoma (CRC) was the second leading cause of cancer death globally. Current literature utilizes reported databases such as Surveillance, Epidemiology, and End Results (SEER) to better understand the epidemiology of CRC. The global cancer observatory’s “Cancer Tomorrow” data visualization tools were used to predict the future incidence and mortality of colorectal cancers until 2030 as a guided tool to look over ways to reduce incidence by controlling risk factors of CRC. The total number of CRC is expected to rise by 2030, with a percent change of 17.3%. The expected percent change in colon cancer is more than rectal cancer (19.8% vs. 11.6%). The estimated number of deaths secondary to CRC is expected to increase in 2030, an estimated percent change of 22.2%. The incidence and mortality rate was higher in men vs. women; however, the gap seems to be closing on trend analysis. Major risk factors for CRC include familial syndromes, family history, race, gender, obesity, diet, alcohol, and smoking. Risk can be reduced by exercise and dietary changes, fiber intake, vitamin D, calcium, and minerals. Individualized screening based on age, gender, and additional risk factors could be an option that needs further comparative data to propose a definitive benefit over established screening guidelines.
According to the Global Cancer Observatory (GLOBOCAN) 2020, colorectal carcinoma (CRC) was the second leading cause of cancer death globally. Current literature utilizes reported databases such as Surveillance, Epidemiology, and End Results (SEER) to better understand the epidemiology of CRC. The global cancer observatory’s “Cancer Tomorrow” data visualization tools were used to predict the future incidence and mortality of colorectal cancers until 2030 as a guided tool to look over ways to reduce incidence by controlling risk factors of CRC. The total number of CRC is expected to rise by 2030, with a percent change of 17.3%. The expected percent change in colon cancer is more than rectal cancer (19.8% vs. 11.6%). The estimated number of deaths secondary to CRC is expected to increase in 2030, an estimated percent change of 22.2%. The incidence and mortality rate was higher in men vs. women; however, the gap seems to be closing on trend analysis. Major risk factors for CRC include familial syndromes, family history, race, gender, obesity, diet, alcohol, and smoking. Risk can be reduced by exercise and dietary changes, fiber intake, vitamin D, calcium, and minerals. Individualized screening based on age, gender, and additional risk factors could be an option that needs further comparative data to propose a definitive benefit over established screening guidelines.
DOI: https://doi.org/10.37349/emed.2021.00063

Antenatal screening for hepatitis B surface antigen seropositivity is widely adopted to identify pregnant women with chronic hepatitis B virus (HBV) infection in order to target their newborn infants for combined passive-active neonatal immunization to prevent the maternal-to-child transmission of HBV. It is less certain whether the presence of chronic HBV infection in these largely asymptomatic women could impact their pregnancy outcome. There is now gathering information in the literature, though sometimes conflicting, on the obstetric implications of chronic HBV infection. The conflicting data is most probably related to confounding factors such as the immunological phase of chronic HBV infection, viral genotype and activity, presence of hepatic inflammation and other co-existing liver disorders such as non-alcoholic fatty liver disease, and coinfection with other virus such as hepatitis C virus and micro-organisms, which are usually not examined, but which could have made significant influence on the occurrence of many of the pregnancy complications and adverse fetal and neonatal outcome. For pregnancy complications, the evidence suggests association with increased gestational diabetes mellitus, preterm birth, intrahepatic cholestasis of pregnancy, caesarean delivery, and postpartum haemorrhage, probably increased placental abruption and prelabour rupture of the membranes, and no effect or a reduction in the hypertensive disorders of pregnancy, especially preeclampsia. For perinatal outcome, there may be increased miscarriage and fetal malformations, and increase in both low birthweight and large-for-gestational age/macrosomic infants, as well as increased intrauterine fetal demise/stillbirth and fetal distress. However, most studies have not elaborated on the mechanisms or explanations of many of the adverse outcomes. Taken together, maternal chronic HBV infection increases the risk of adverse obstetric outcome overall, but further prospective studies are warranted to elucidate the reasons and mechanisms of, and with a view to mitigating, these adverse obstetric outcomes.
Antenatal screening for hepatitis B surface antigen seropositivity is widely adopted to identify pregnant women with chronic hepatitis B virus (HBV) infection in order to target their newborn infants for combined passive-active neonatal immunization to prevent the maternal-to-child transmission of HBV. It is less certain whether the presence of chronic HBV infection in these largely asymptomatic women could impact their pregnancy outcome. There is now gathering information in the literature, though sometimes conflicting, on the obstetric implications of chronic HBV infection. The conflicting data is most probably related to confounding factors such as the immunological phase of chronic HBV infection, viral genotype and activity, presence of hepatic inflammation and other co-existing liver disorders such as non-alcoholic fatty liver disease, and coinfection with other virus such as hepatitis C virus and micro-organisms, which are usually not examined, but which could have made significant influence on the occurrence of many of the pregnancy complications and adverse fetal and neonatal outcome. For pregnancy complications, the evidence suggests association with increased gestational diabetes mellitus, preterm birth, intrahepatic cholestasis of pregnancy, caesarean delivery, and postpartum haemorrhage, probably increased placental abruption and prelabour rupture of the membranes, and no effect or a reduction in the hypertensive disorders of pregnancy, especially preeclampsia. For perinatal outcome, there may be increased miscarriage and fetal malformations, and increase in both low birthweight and large-for-gestational age/macrosomic infants, as well as increased intrauterine fetal demise/stillbirth and fetal distress. However, most studies have not elaborated on the mechanisms or explanations of many of the adverse outcomes. Taken together, maternal chronic HBV infection increases the risk of adverse obstetric outcome overall, but further prospective studies are warranted to elucidate the reasons and mechanisms of, and with a view to mitigating, these adverse obstetric outcomes.
DOI: https://doi.org/10.37349/emed.2021.00064

Aim:
The high heterogeneity in the definitions of low back pain encountered in the literature has led to the development of standardized definitions of this condition called “Delphi definitions of low back pain prevalence (Delphi DOLBaPP)” by a group of international researchers. In order to be widely used, these definitions need to be adapted according to the cultural and linguistic context. The aim of this work was to perform the cross-cultural adaptation of the Delphi DOLBaPP definitions in Quebec French and to pre-test them among French-speaking adults.
Methods:
In order to enable practical use of the Delphi DOLBaPP definitions in different contexts, their presentation was adapted in the form of a questionnaire (referred to as the “Delphi DOLBaPP questionnaire”). The process of cross-cultural adaptation of the Delphi DOLBaPP questionnaire in French was conducted according to the most recognized recommendations for the cultural adaptation of measuring instruments. The resulting questionnaire and an evaluation form were then submitted to a sample of 82 adults.
Results:
A total of 41 participants (50.0%) reported low back pain. A high proportion of participants (89.0%) stated that it took them less than 5 minutes to complete the questionnaire. More than 62.0% of them did not find any question poorly worded or confusing. Nearly 80.0% of the participants found the questionnaire easy to understand. The cross-cultural adaptation process suggested minor modifications to the original Delphi DOLBaPP questionnaire.
Conclusions:
This study has produced a cross-cultural adaptation of the Delphi DOLBaPP questionnaire in Quebec French that will enable French-speaking populations to share the benefits of using standardized definitions of low back pain in epidemiological studies.
Aim:
The high heterogeneity in the definitions of low back pain encountered in the literature has led to the development of standardized definitions of this condition called “Delphi definitions of low back pain prevalence (Delphi DOLBaPP)” by a group of international researchers. In order to be widely used, these definitions need to be adapted according to the cultural and linguistic context. The aim of this work was to perform the cross-cultural adaptation of the Delphi DOLBaPP definitions in Quebec French and to pre-test them among French-speaking adults.
Methods:
In order to enable practical use of the Delphi DOLBaPP definitions in different contexts, their presentation was adapted in the form of a questionnaire (referred to as the “Delphi DOLBaPP questionnaire”). The process of cross-cultural adaptation of the Delphi DOLBaPP questionnaire in French was conducted according to the most recognized recommendations for the cultural adaptation of measuring instruments. The resulting questionnaire and an evaluation form were then submitted to a sample of 82 adults.
Results:
A total of 41 participants (50.0%) reported low back pain. A high proportion of participants (89.0%) stated that it took them less than 5 minutes to complete the questionnaire. More than 62.0% of them did not find any question poorly worded or confusing. Nearly 80.0% of the participants found the questionnaire easy to understand. The cross-cultural adaptation process suggested minor modifications to the original Delphi DOLBaPP questionnaire.
Conclusions:
This study has produced a cross-cultural adaptation of the Delphi DOLBaPP questionnaire in Quebec French that will enable French-speaking populations to share the benefits of using standardized definitions of low back pain in epidemiological studies.
DOI: https://doi.org/10.37349/emed.2021.00065

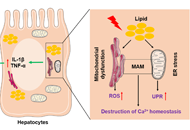
Non-alcoholic fatty liver disease (NAFLD) as the most common chronic liver disease poses a significant impact on public healthcare and economic risk worldwide. As a multifactorial disease, NAFLD is usually associated with many comorbidities such as obesity, insulin resistance, hypertension, hyperlipidemia, diabetes, and cardiovascular disease. Without effectively preventive intervention, the advanced stage of NAFLD, non-alcoholic steatohepatitis (NASH), can progress to cirrhosis and hepatocellular carcinoma (HCC). However, there is no approved therapeutic treatment. Excessive fat accumulation in the liver is the hallmark of NAFLD, which can lead to mitochondrial dysfunction and endoplasmic reticulum (ER) stress. Dysfunction of two organelles also induces the upregulation of reactive oxygen species (ROS), activation of the unfolded protein response (UPR), and disruption of calcium transport, which promote NAFLD progression. Herein, this review summarized the current understanding of the roles of mitochondrial dysfunction and ER stress in the pathogenesis of NAFLD. Specifically, this review focused on the key molecules associated with the ER-mitochondria communication and different treatment options by targeting ER stress and mitochondrial dysfunction to treat NAFLD or NASH. Clinical trials to evaluate the therapeutic efficacy of representative agents, such as natural products, metabolites, and modulators of stress, have been reviewed and analyzed. Overall, recent findings suggest that targeting ER stress and mitochondrial dysfunction holds a promise for NAFLD treatment.
Non-alcoholic fatty liver disease (NAFLD) as the most common chronic liver disease poses a significant impact on public healthcare and economic risk worldwide. As a multifactorial disease, NAFLD is usually associated with many comorbidities such as obesity, insulin resistance, hypertension, hyperlipidemia, diabetes, and cardiovascular disease. Without effectively preventive intervention, the advanced stage of NAFLD, non-alcoholic steatohepatitis (NASH), can progress to cirrhosis and hepatocellular carcinoma (HCC). However, there is no approved therapeutic treatment. Excessive fat accumulation in the liver is the hallmark of NAFLD, which can lead to mitochondrial dysfunction and endoplasmic reticulum (ER) stress. Dysfunction of two organelles also induces the upregulation of reactive oxygen species (ROS), activation of the unfolded protein response (UPR), and disruption of calcium transport, which promote NAFLD progression. Herein, this review summarized the current understanding of the roles of mitochondrial dysfunction and ER stress in the pathogenesis of NAFLD. Specifically, this review focused on the key molecules associated with the ER-mitochondria communication and different treatment options by targeting ER stress and mitochondrial dysfunction to treat NAFLD or NASH. Clinical trials to evaluate the therapeutic efficacy of representative agents, such as natural products, metabolites, and modulators of stress, have been reviewed and analyzed. Overall, recent findings suggest that targeting ER stress and mitochondrial dysfunction holds a promise for NAFLD treatment.
DOI: https://doi.org/10.37349/emed.2021.00066
This article belongs to the special issue Exploring Chronic Liver Disease

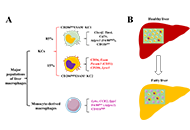
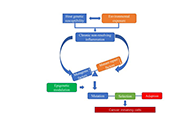
Tissue-resident macrophages play critically important roles in host homeostasis and pathogenesis of diseases, with the functions of phagocytosis, metabolism, and immune modulation. Recently, two research studies accomplished by a collaborated group of researchers showed that there are two populations of liver resident Kupffer cells (KCs), including a major cluster of differentiation 206 low expression (CD206low)endothelial cell-selective adhesion molecule negative (ESAM−) population (KC1) and a minor CD206highESAM+ population (KC2). Both KC1 and KC2 express KC markers, such as C-type lectin domain family 4 member F (CLEC4F) and T-cell membrane protein 4 (Tim4). In fatty liver, the frequency of KC2 was increased, and those KC2 expressed some markers like liver sinusoidal endothelial cells (LSECs), such as CD31 and ESAM. In addition, KC2 population had a relatively higher expression of CD36, as fatty acid transporter, which was implicated in the production of reactive oxygen species (ROS) and lipid peroxidation. Furthermore, this collaborated group also showed that KC2 can cross-present hepatocellular antigens to prime antiviral function of CD8+ T cells by sensing interleukin-2 (IL-2) in hepatitis B virus (HBV) replication-competent transgenic mice. Increasing evidence shows that targeting hepatic macrophages can prevent and reverse non-alcoholic fatty liver disease (NAFLD), with a new suggested name metabolic dysfunction-associated fatty liver disease (MAFLD) to include metabolic dysfunction-associated fatty liver diseases, such as viruses and alcohol. In summary, differentiating specific populations of hepatic macrophages is critically important for the treatment of MAFLD or NAFLD, and their overlaps. Markers specifically expressed on sub-types of hepatic macrophages may be applied for liver disease diagnosis.
Tissue-resident macrophages play critically important roles in host homeostasis and pathogenesis of diseases, with the functions of phagocytosis, metabolism, and immune modulation. Recently, two research studies accomplished by a collaborated group of researchers showed that there are two populations of liver resident Kupffer cells (KCs), including a major cluster of differentiation 206 low expression (CD206low)endothelial cell-selective adhesion molecule negative (ESAM−) population (KC1) and a minor CD206highESAM+ population (KC2). Both KC1 and KC2 express KC markers, such as C-type lectin domain family 4 member F (CLEC4F) and T-cell membrane protein 4 (Tim4). In fatty liver, the frequency of KC2 was increased, and those KC2 expressed some markers like liver sinusoidal endothelial cells (LSECs), such as CD31 and ESAM. In addition, KC2 population had a relatively higher expression of CD36, as fatty acid transporter, which was implicated in the production of reactive oxygen species (ROS) and lipid peroxidation. Furthermore, this collaborated group also showed that KC2 can cross-present hepatocellular antigens to prime antiviral function of CD8+ T cells by sensing interleukin-2 (IL-2) in hepatitis B virus (HBV) replication-competent transgenic mice. Increasing evidence shows that targeting hepatic macrophages can prevent and reverse non-alcoholic fatty liver disease (NAFLD), with a new suggested name metabolic dysfunction-associated fatty liver disease (MAFLD) to include metabolic dysfunction-associated fatty liver diseases, such as viruses and alcohol. In summary, differentiating specific populations of hepatic macrophages is critically important for the treatment of MAFLD or NAFLD, and their overlaps. Markers specifically expressed on sub-types of hepatic macrophages may be applied for liver disease diagnosis.
DOI: https://doi.org/10.37349/emed.2021.00067
This article belongs to the special issue Exploring Chronic Liver Disease

The processes of cancer and embryonic development have a partially overlapping effect. Several transcription factor families, which are highly conserved in the evolutionary history of biology, play a key role in the development of cancer and are often responsible for the pivotal developmental processes such as cell survival, expansion, senescence, and differentiation. As an evolutionary conserved and ubiquitously expression protein, CCCTC-binding factor (CTCF) has diverse regulatory functions, including gene regulation, imprinting, insulation, X chromosome inactivation, and the establishment of three-dimensional (3D) chromatin structure during human embryogenesis. In various cancers, CTCF is considered as a tumor suppressor gene and plays homeostatic roles in maintaining genome function and integrity. However, the mechanisms of CTCF in tumor development have not been fully elucidated. Here, this review will focus on the key roles of CTCF in cancer evolution and development (Cancer Evo-Dev) and embryogenesis.
The processes of cancer and embryonic development have a partially overlapping effect. Several transcription factor families, which are highly conserved in the evolutionary history of biology, play a key role in the development of cancer and are often responsible for the pivotal developmental processes such as cell survival, expansion, senescence, and differentiation. As an evolutionary conserved and ubiquitously expression protein, CCCTC-binding factor (CTCF) has diverse regulatory functions, including gene regulation, imprinting, insulation, X chromosome inactivation, and the establishment of three-dimensional (3D) chromatin structure during human embryogenesis. In various cancers, CTCF is considered as a tumor suppressor gene and plays homeostatic roles in maintaining genome function and integrity. However, the mechanisms of CTCF in tumor development have not been fully elucidated. Here, this review will focus on the key roles of CTCF in cancer evolution and development (Cancer Evo-Dev) and embryogenesis.
DOI: https://doi.org/10.37349/emed.2021.00068

Pseudoexfoliation (PXF) syndrome is an important public health concern requiring individual population level analysis. Disease prevalence differs by geographic location and ethnicity, and has environmental, demographic, genetic, and molecular risk factors have been demonstrated. Epidemiological factors that have been associated with PXF include age, sex, environmental factors, and diet. Genetic and molecular components have also been identified that are associated with PXF. Underserved populations are often understudied within scientific research, including research about eye disease such as PXF, contributing to the persistence of health disparities within these populations. In each population, PXF needs may be different, and by having research that identifies individual population needs about PXF, the resources in that population can be more efficiently utilized. Otherwise, PXF intervention and care management based only on the broadest level of understanding may continue to exacerbate health disparities in populations disproportionally burdened by PXF.
Pseudoexfoliation (PXF) syndrome is an important public health concern requiring individual population level analysis. Disease prevalence differs by geographic location and ethnicity, and has environmental, demographic, genetic, and molecular risk factors have been demonstrated. Epidemiological factors that have been associated with PXF include age, sex, environmental factors, and diet. Genetic and molecular components have also been identified that are associated with PXF. Underserved populations are often understudied within scientific research, including research about eye disease such as PXF, contributing to the persistence of health disparities within these populations. In each population, PXF needs may be different, and by having research that identifies individual population needs about PXF, the resources in that population can be more efficiently utilized. Otherwise, PXF intervention and care management based only on the broadest level of understanding may continue to exacerbate health disparities in populations disproportionally burdened by PXF.
DOI: https://doi.org/10.37349/emed.2021.00069

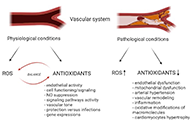
Reactive oxygen species (ROS) and reactive nitrogen species (RNS) play a fundamental role in regulating endothelial function and vascular tone in the physiological conditions of a vascular system. However, oxidative stress has detrimental effects on human health, and numerous studies confirmed that high ROS/RNS production contributes to the initiation and progression of cardiovascular diseases. The antioxidant defense has an essential role in the homeostatic functioning of the vascular endothelial system. Endogenous antioxidative defense includes various molecules and enzymes such as superoxide dismutase, catalase, glutathione reductase, and glutathione peroxidase. Together all these antioxidative enzymes are essential for defense against harmful ROS features. ROS are mainly generated from redox-active compounds involved in the mitochondrial respiratory chain. Thus, targeting antioxidative enzymes and mitochondria oxidative balance may be a promising approach for vascular diseases occurrence and treatment. This review summarized the most recent research on the regulation of antioxidative enzymes in vascular diseases.
Reactive oxygen species (ROS) and reactive nitrogen species (RNS) play a fundamental role in regulating endothelial function and vascular tone in the physiological conditions of a vascular system. However, oxidative stress has detrimental effects on human health, and numerous studies confirmed that high ROS/RNS production contributes to the initiation and progression of cardiovascular diseases. The antioxidant defense has an essential role in the homeostatic functioning of the vascular endothelial system. Endogenous antioxidative defense includes various molecules and enzymes such as superoxide dismutase, catalase, glutathione reductase, and glutathione peroxidase. Together all these antioxidative enzymes are essential for defense against harmful ROS features. ROS are mainly generated from redox-active compounds involved in the mitochondrial respiratory chain. Thus, targeting antioxidative enzymes and mitochondria oxidative balance may be a promising approach for vascular diseases occurrence and treatment. This review summarized the most recent research on the regulation of antioxidative enzymes in vascular diseases.
DOI: https://doi.org/10.37349/emed.2021.00070
This article belongs to the special issue Reactive Oxygen Species (ROS) in Pathophysiological Conditions

Aim:
Isolated specific glycone–aglycone conjugated flavonoids which are investigated for their effect of bioavailability and molecular concentrations. The specific formula is then tested via in vitro and in vivo cytotoxicity tests.
Methods:
Considering the higher affinity for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), quercetin, quercetin 3-sambubioside-3’-glucoside, luteolin, apigenin-7-4’alloside, kaempferol-7-O-glucoside, epicatechin-epigallocatechin-3-O-gallate, and hesperetin were selected to investigate the effects of a new combination of the formula. Specific chemical analyses, such as high-performance liquid chromatography (HPLC), liquid chromatography–mass spectrometry (LC–MS), quadrupole time of flight mass spectrometry (QTOF–MS) analysis and ultraviolet–visible (UV–VIS) spectrophotometry, were performed for molecular qualification and quantification.
Results:
In silico molecular docking analyses have shown that flavonoids can bind strongly to the spike protein and main protease of the SARS-CoV-2 virus. Flavonoids also have anti-inflammatory and immune-modulating activity by inhibiting cytokines. Although flavonoids may be a treatment alternative for coronavirus disease 2019 (COVID-19), an effective flavonoid compound has yet to be developed. The main problem here is that the absorption rate of flavonoids is very low (2–10%) in the intestines, and these compounds are metabolized rapidly. In contrast, according to recent literature, a conjugated flavonoid mixture is better absorbed in the small intestine, and its toxic effects are relatively fewer.
Conclusions:
It is found that the new formula has no cytotoxic or genotoxic effects. Furthermore, oral administrations of the new compound did not produce any toxicity symptoms or any mortality in male and female rats. The pre-clinical in vitro and in vivo toxicity test results indicated that the new flavonoid formula can be safely used for clinical trials.
Aim:
Isolated specific glycone–aglycone conjugated flavonoids which are investigated for their effect of bioavailability and molecular concentrations. The specific formula is then tested via in vitro and in vivo cytotoxicity tests.
Methods:
Considering the higher affinity for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), quercetin, quercetin 3-sambubioside-3’-glucoside, luteolin, apigenin-7-4’alloside, kaempferol-7-O-glucoside, epicatechin-epigallocatechin-3-O-gallate, and hesperetin were selected to investigate the effects of a new combination of the formula. Specific chemical analyses, such as high-performance liquid chromatography (HPLC), liquid chromatography–mass spectrometry (LC–MS), quadrupole time of flight mass spectrometry (QTOF–MS) analysis and ultraviolet–visible (UV–VIS) spectrophotometry, were performed for molecular qualification and quantification.
Results:
In silico molecular docking analyses have shown that flavonoids can bind strongly to the spike protein and main protease of the SARS-CoV-2 virus. Flavonoids also have anti-inflammatory and immune-modulating activity by inhibiting cytokines. Although flavonoids may be a treatment alternative for coronavirus disease 2019 (COVID-19), an effective flavonoid compound has yet to be developed. The main problem here is that the absorption rate of flavonoids is very low (2–10%) in the intestines, and these compounds are metabolized rapidly. In contrast, according to recent literature, a conjugated flavonoid mixture is better absorbed in the small intestine, and its toxic effects are relatively fewer.
Conclusions:
It is found that the new formula has no cytotoxic or genotoxic effects. Furthermore, oral administrations of the new compound did not produce any toxicity symptoms or any mortality in male and female rats. The pre-clinical in vitro and in vivo toxicity test results indicated that the new flavonoid formula can be safely used for clinical trials.
DOI: https://doi.org/10.37349/emed.2022.00071

Cancer is the leading cause of mortality worldwide, which necessitates our consideration related to novel treatment approach. Tumor cells at the tumor microenvironment (TME), regulate a plethora of key mechanistic signaling pathways that obstruct antitumor immune responses by immune suppression, immune resistance or acquired immune tolerance. The present therapeutic regimes are provided independently or in combination, or as immunotherapies for cancer immune targeting. Immunotherapy has altered the arena of oncology and patient care. By using the host immune system, the immunostimulatory molecules can exert a robust, personalized response against the patient’s own tumors. Alternatively, tumors may exploit these strategies to escape immune recognition, and accordingly, such mechanisms represent chances for immunotherapy intervention. Nonetheless, despite promising outcomes from immunotherapies in recurrent and metastatic cancers, immune-therapeutics in clinics have been limited owing to unpredictability in the produced immune response and reported instances of immune-related adverse effects. The unrealized potential of immunotherapies in cancer management maybe due to the obstacles such as heterogeneous nature, multiple targets, patients’ immune response, specificity for cancer or variability in response generation in toxicity levels, delivery and cost related to therapeutics etc. Further revolutionary trends related to immunotherapies are noticeable with slower progress for cancer management. Recent advances in nanomedicine strategize to ameliorate the lacuna of immunotherapy as it relies on the inherent biophysical characteristics of nanocarriers: size, shape, surface charge and multifunctionality and exploiting them as first line therapy for delivery of biomolecules, single checkpoint inhibitors and for imaging of TME. Therefore, nano-assisted immunotherapies can boost the immunotherapeutic approach, overcoming factors that are with imminent potential risks related to it, thereby significantly improving the survival rate associated with it in cancer patients. Nanotechnology is anticipated to overcome the confines of existing cancer immunotherapy and to successfully combine various cancer treatment modes.
Cancer is the leading cause of mortality worldwide, which necessitates our consideration related to novel treatment approach. Tumor cells at the tumor microenvironment (TME), regulate a plethora of key mechanistic signaling pathways that obstruct antitumor immune responses by immune suppression, immune resistance or acquired immune tolerance. The present therapeutic regimes are provided independently or in combination, or as immunotherapies for cancer immune targeting. Immunotherapy has altered the arena of oncology and patient care. By using the host immune system, the immunostimulatory molecules can exert a robust, personalized response against the patient’s own tumors. Alternatively, tumors may exploit these strategies to escape immune recognition, and accordingly, such mechanisms represent chances for immunotherapy intervention. Nonetheless, despite promising outcomes from immunotherapies in recurrent and metastatic cancers, immune-therapeutics in clinics have been limited owing to unpredictability in the produced immune response and reported instances of immune-related adverse effects. The unrealized potential of immunotherapies in cancer management maybe due to the obstacles such as heterogeneous nature, multiple targets, patients’ immune response, specificity for cancer or variability in response generation in toxicity levels, delivery and cost related to therapeutics etc. Further revolutionary trends related to immunotherapies are noticeable with slower progress for cancer management. Recent advances in nanomedicine strategize to ameliorate the lacuna of immunotherapy as it relies on the inherent biophysical characteristics of nanocarriers: size, shape, surface charge and multifunctionality and exploiting them as first line therapy for delivery of biomolecules, single checkpoint inhibitors and for imaging of TME. Therefore, nano-assisted immunotherapies can boost the immunotherapeutic approach, overcoming factors that are with imminent potential risks related to it, thereby significantly improving the survival rate associated with it in cancer patients. Nanotechnology is anticipated to overcome the confines of existing cancer immunotherapy and to successfully combine various cancer treatment modes.
DOI: https://doi.org/10.37349/emed.2022.00072
This article belongs to the special issue Nanomedicine and Cancer Immunotherapy

The redox status in pathogenesis is critically regulated by careful balance between the generation of reactive oxygen species (ROS) and their elimination. Increased ROS level above the cellular tolerability threshold results in apoptotic or necrotic cell death. ROS belongs to a group of highly reactive compounds that have evolved to play key roles in cellular signaling pathways. It’s widely assumed that a reasonable amount of ROS is essential for a variety of biological processes. Elevated levels of ROS are known to cause various pathologic conditions like neurological disorders, cardiovascular conditions, inflammation, autoimmunity, and cancer. ROS is well known to initiate and assist in progression of tumor by promoting proliferation and survival of cancer cells and thus facilitates pro-tumorigenic signaling in tumor microenvironment. As cancer cells become more resilient to the effects of ROS manipulating drugs, increased antioxidant capacity attenuates their susceptibility to cancer treatment. Excessive environmental stress, on the other hand, can cause cancer cells to die. This review summarizes various molecular mechanisms including the role of checkpoint inhibitors that can be harnessed to develop effective therapeutic strategies for targeting ROS related signaling in cancer.
The redox status in pathogenesis is critically regulated by careful balance between the generation of reactive oxygen species (ROS) and their elimination. Increased ROS level above the cellular tolerability threshold results in apoptotic or necrotic cell death. ROS belongs to a group of highly reactive compounds that have evolved to play key roles in cellular signaling pathways. It’s widely assumed that a reasonable amount of ROS is essential for a variety of biological processes. Elevated levels of ROS are known to cause various pathologic conditions like neurological disorders, cardiovascular conditions, inflammation, autoimmunity, and cancer. ROS is well known to initiate and assist in progression of tumor by promoting proliferation and survival of cancer cells and thus facilitates pro-tumorigenic signaling in tumor microenvironment. As cancer cells become more resilient to the effects of ROS manipulating drugs, increased antioxidant capacity attenuates their susceptibility to cancer treatment. Excessive environmental stress, on the other hand, can cause cancer cells to die. This review summarizes various molecular mechanisms including the role of checkpoint inhibitors that can be harnessed to develop effective therapeutic strategies for targeting ROS related signaling in cancer.
DOI: https://doi.org/10.37349/emed.2022.00073
This article belongs to the special issue Reactive Oxygen Species (ROS) in Pathophysiological Conditions

Since obesity is one of the main factors in the development of insulin resistance (IR) and is also associated with increased oxidative stress (OxS) rate, this study aims to review the published literature to collate and provide a comprehensive summary of the studies related to the status of the OxS in the pathogenesis of obesity and related IR. OxS represents an imbalance between the production of reactive oxygen and nitrogen species (RONS) and the capacity of the antioxidant defense system (AOS) to neutralize RONS. A steady-state of RONS level is maintained through endogenous enzymatic and non-enzymatic AOS components. Three crucial enzymes, which suppress the formation of free radicals, are superoxide dismutases, catalases, and glutathione peroxidases. The second line of AOS includes non-enzymatic components such as vitamins C and E, coenzyme Q, and glutathione which neutralizes free radicals by donating electrons to RONS. Emerging evidence suggests that high RONS levels contribute to the progression of OxS in obesity by activating inflammatory pathways and thus leading to the development of pathological states, including IR. In addition, decreased level of AOS components in obesity increases the susceptibility to oxidative tissue damage and further progression of its comorbidities. Increased OxS in accumulated adipose tissue should be an imperative target for developing new therapies in obesity-related IR.
Since obesity is one of the main factors in the development of insulin resistance (IR) and is also associated with increased oxidative stress (OxS) rate, this study aims to review the published literature to collate and provide a comprehensive summary of the studies related to the status of the OxS in the pathogenesis of obesity and related IR. OxS represents an imbalance between the production of reactive oxygen and nitrogen species (RONS) and the capacity of the antioxidant defense system (AOS) to neutralize RONS. A steady-state of RONS level is maintained through endogenous enzymatic and non-enzymatic AOS components. Three crucial enzymes, which suppress the formation of free radicals, are superoxide dismutases, catalases, and glutathione peroxidases. The second line of AOS includes non-enzymatic components such as vitamins C and E, coenzyme Q, and glutathione which neutralizes free radicals by donating electrons to RONS. Emerging evidence suggests that high RONS levels contribute to the progression of OxS in obesity by activating inflammatory pathways and thus leading to the development of pathological states, including IR. In addition, decreased level of AOS components in obesity increases the susceptibility to oxidative tissue damage and further progression of its comorbidities. Increased OxS in accumulated adipose tissue should be an imperative target for developing new therapies in obesity-related IR.
DOI: https://doi.org/10.37349/emed.2022.00074
This article belongs to the special issue Reactive Oxygen Species (ROS) in Pathophysiological Conditions

DOI: https://doi.org/10.37349/emed.2022.00075
This article belongs to the special issue Exploring Chronic Liver Disease

T cells play a central role in anti-tumor immunity, and reactive oxygen species (ROS) lie at the crossroad on the anti-tumor T cell responses. To activate efficient T cell immunity, a moderate level of ROS is needed, however, excessive ROS would cause toxicity to the T cells, because the improper level leads to the formation and maintenance of an immunosuppressive tumor microenvironment. Up to date, strategies that modulate ROS, either increasing or decreasing, have been widely investigated. Some of them are utilized in anti-tumor therapies, showing inevitable impacts on the anti-tumor T cell immunity with both obverse and reverse sides. Herein, the impacts of ROS-increasing and ROS-decreasing treatments on the T cell responses in the tumor microenvironment are reviewed and discussed. At the same time, outcomes of combination immunotherapies are introduced to put forward inspirations to unleash the potential of immunotherapies.
T cells play a central role in anti-tumor immunity, and reactive oxygen species (ROS) lie at the crossroad on the anti-tumor T cell responses. To activate efficient T cell immunity, a moderate level of ROS is needed, however, excessive ROS would cause toxicity to the T cells, because the improper level leads to the formation and maintenance of an immunosuppressive tumor microenvironment. Up to date, strategies that modulate ROS, either increasing or decreasing, have been widely investigated. Some of them are utilized in anti-tumor therapies, showing inevitable impacts on the anti-tumor T cell immunity with both obverse and reverse sides. Herein, the impacts of ROS-increasing and ROS-decreasing treatments on the T cell responses in the tumor microenvironment are reviewed and discussed. At the same time, outcomes of combination immunotherapies are introduced to put forward inspirations to unleash the potential of immunotherapies.
DOI: https://doi.org/10.37349/emed.2022.00076
This article belongs to the special issue Reactive Oxygen Species (ROS) in Pathophysiological Conditions

Aim:
Preeclampsia (PE) is a hypertensive disorder of pregnancy associated with activated CD4+ T cells and autoantibodies to angiotensin II type 1 receptor (AT1-AA). We have previously shown that CD4+ T cells isolated from women with PE cause hypertension, increased tumor necrosis factor alpha (TNF-α), endothelin-1, and soluble fms-like tyrosine kinase-1 (sFlt-1) when injected into pregnant nude-athymic rats compared to CD4+ T cells from normal pregnant (NP) women. However, the role of PE CD4+ T cells to cause AT1-AA as a mechanism of hypertension is not known. Our goal was to determine if PE CD4+ T cells stimulate AT1-AA in pregnant nude-athymic rats.
Methods:
CD4+ T cells were isolated from human NP and PE placentasand injected into nude-athymic rats on gestational day (GD) 12. In order to examine the role of the PE CD4+ T cells to stimulate B cell secretion of AT1-AA, a subset of the rats receiving PE CD4+ T cells were treated with rituximab on GD 14 or anti-CD40 ligand (anti-CD40L) on GD 12. On GD 19, mean arterial pressure (MAP) and tissues were obtained.
Results:
MAP [114 ± 1 mmHg (n = 9)] and AT1-AA [19.8 ± 0.9 beats per minute (bpm, n = 4)] were increased in NP nude + PE CD4+ T cells compared to NP nude + NP CD4+ T cells [98 ± 2 mmHg (n = 7, P < 0.05) and 1.3 ± 0.9 bpm (n = 5, P < 0.05)]. Rituximab (103 ± 2 mmHg, n = 3, P < 0.05) and anti-CD40L (102 ± 1 mmHg, n = 3, P < 0.05) lowered MAP compared to NP nude + PE CD4+ T cells. Circulating a proliferation-inducing ligand (APRIL) and placental angiotensin-converting enzyme 2 (ACE-2) activity was increased in response to PE CD4+ T cells.
Conclusions:
These results show that placental CD4+ T cells play an important role in the pathophysiology of PE, by activating B cells secreting AT1-AA to cause hypertension during pregnancy.
Aim:
Preeclampsia (PE) is a hypertensive disorder of pregnancy associated with activated CD4+ T cells and autoantibodies to angiotensin II type 1 receptor (AT1-AA). We have previously shown that CD4+ T cells isolated from women with PE cause hypertension, increased tumor necrosis factor alpha (TNF-α), endothelin-1, and soluble fms-like tyrosine kinase-1 (sFlt-1) when injected into pregnant nude-athymic rats compared to CD4+ T cells from normal pregnant (NP) women. However, the role of PE CD4+ T cells to cause AT1-AA as a mechanism of hypertension is not known. Our goal was to determine if PE CD4+ T cells stimulate AT1-AA in pregnant nude-athymic rats.
Methods:
CD4+ T cells were isolated from human NP and PE placentasand injected into nude-athymic rats on gestational day (GD) 12. In order to examine the role of the PE CD4+ T cells to stimulate B cell secretion of AT1-AA, a subset of the rats receiving PE CD4+ T cells were treated with rituximab on GD 14 or anti-CD40 ligand (anti-CD40L) on GD 12. On GD 19, mean arterial pressure (MAP) and tissues were obtained.
Results:
MAP [114 ± 1 mmHg (n = 9)] and AT1-AA [19.8 ± 0.9 beats per minute (bpm, n = 4)] were increased in NP nude + PE CD4+ T cells compared to NP nude + NP CD4+ T cells [98 ± 2 mmHg (n = 7, P < 0.05) and 1.3 ± 0.9 bpm (n = 5, P < 0.05)]. Rituximab (103 ± 2 mmHg, n = 3, P < 0.05) and anti-CD40L (102 ± 1 mmHg, n = 3, P < 0.05) lowered MAP compared to NP nude + PE CD4+ T cells. Circulating a proliferation-inducing ligand (APRIL) and placental angiotensin-converting enzyme 2 (ACE-2) activity was increased in response to PE CD4+ T cells.
Conclusions:
These results show that placental CD4+ T cells play an important role in the pathophysiology of PE, by activating B cells secreting AT1-AA to cause hypertension during pregnancy.
DOI: https://doi.org/10.37349/emed.2022.00077
This article belongs to the special issue Angiotensins—A Century of Progress

Melatonin is the primary hormone of the pineal gland that is secreted at night. It regulates many physiological functions, including the sleep-wake cycle, gonadal activity, free radical scavenging, immunomodulation, neuro-protection, and cancer progression. The precise functions of melatonin are mediated by guanosine triphosphate (GTP)-binding protein (G-protein) coupled melatonin receptor 1 (MT1) and MT2 receptors. However, nuclear receptors are also associated with melatonin activity. Circadian rhythm disruption, shift work, and light exposure at night hamper melatonin production. Impaired melatonin level promotes various pathophysiological changes, including cancer. In our modern society, breast cancer is a serious problem throughout the world. Several studies have been indicated the link between low levels of melatonin and breast cancer development. Melatonin has oncostatic properties in breast cancer cells. This indolamine advances apoptosis, which arrests the cell cycle and regulates metabolic activity. Moreover, melatonin increases the treatment efficacy of cancer and can be used as an adjuvant with chemotherapeutic agents.
Melatonin is the primary hormone of the pineal gland that is secreted at night. It regulates many physiological functions, including the sleep-wake cycle, gonadal activity, free radical scavenging, immunomodulation, neuro-protection, and cancer progression. The precise functions of melatonin are mediated by guanosine triphosphate (GTP)-binding protein (G-protein) coupled melatonin receptor 1 (MT1) and MT2 receptors. However, nuclear receptors are also associated with melatonin activity. Circadian rhythm disruption, shift work, and light exposure at night hamper melatonin production. Impaired melatonin level promotes various pathophysiological changes, including cancer. In our modern society, breast cancer is a serious problem throughout the world. Several studies have been indicated the link between low levels of melatonin and breast cancer development. Melatonin has oncostatic properties in breast cancer cells. This indolamine advances apoptosis, which arrests the cell cycle and regulates metabolic activity. Moreover, melatonin increases the treatment efficacy of cancer and can be used as an adjuvant with chemotherapeutic agents.
DOI: https://doi.org/10.37349/emed.2022.00078

Aim:
The Losartan Intervention For Endpoint reduction in hypertension (LIFE) study showed less new-onset atrial fibrillation (AF) in hypertensive patients receiving losartan- vs. atenolol-based treatment. Because losartan reduces serum uric acid (SUA) levels, the aim of the present study was to investigate relations of SUA with new-onset AF in the study.
Methods:
Hypertensive patients with electrocardiographic (ECG) left ventricular hypertrophy (LVH) and no prior AF (n = 8,243) were treated for 5.0 ± 0.4 years with losartan- or atenolol-based therapy. Associations of SUA with new-onset AF documented by Minnesota coding were assessed by Cox models using SUA and systolic blood pressure as time-varying covariates to take into account changes of SUA related to losartan or diuretic treatment, changes in renal function, and aging.
Results:
Time-varying SUA was associated with new AF defined by Minnesota code [hazard ratio (HR) = 1.19 per 16.8 μmol/L (1 mg/dL), (95% confidence intervals (CIs), 1.12–1.26), P < 0.0001], independent of losartan treatment [HR = 0.75 (95% CIs, 0.61–0.93), P = 0.007], older age [HR = 1.95 per 7.0 years (95% CIs, 1.73–2.20), P < 0.0001], male sex [HR = 1.46 (95% CIs, 1.09–1.94), P = 0.010] and higher Cornell voltage-duration product [HR = 1.10 per 1,023 ms·mm (95% CIs, 1.01–1.21), P = 0.034]. Similar results were obtained in Cox models with SUA levels partitioned according to baseline quartiles and in which AF was defined by physician reports or by both Minnesota coding and physician reports.
Conclusions:
In-treatment SUA is a strong predictor for new-onset AF in hypertensive patients, independent of effects of antihypertensive treatment, age, sex, and ECG-LVH. Further research is needed to clarify how uric acid may provoke AF (ClinicalTrials.gov identifier: NCT00338260).
Aim:
The Losartan Intervention For Endpoint reduction in hypertension (LIFE) study showed less new-onset atrial fibrillation (AF) in hypertensive patients receiving losartan- vs. atenolol-based treatment. Because losartan reduces serum uric acid (SUA) levels, the aim of the present study was to investigate relations of SUA with new-onset AF in the study.
Methods:
Hypertensive patients with electrocardiographic (ECG) left ventricular hypertrophy (LVH) and no prior AF (n = 8,243) were treated for 5.0 ± 0.4 years with losartan- or atenolol-based therapy. Associations of SUA with new-onset AF documented by Minnesota coding were assessed by Cox models using SUA and systolic blood pressure as time-varying covariates to take into account changes of SUA related to losartan or diuretic treatment, changes in renal function, and aging.
Results:
Time-varying SUA was associated with new AF defined by Minnesota code [hazard ratio (HR) = 1.19 per 16.8 μmol/L (1 mg/dL), (95% confidence intervals (CIs), 1.12–1.26), P < 0.0001], independent of losartan treatment [HR = 0.75 (95% CIs, 0.61–0.93), P = 0.007], older age [HR = 1.95 per 7.0 years (95% CIs, 1.73–2.20), P < 0.0001], male sex [HR = 1.46 (95% CIs, 1.09–1.94), P = 0.010] and higher Cornell voltage-duration product [HR = 1.10 per 1,023 ms·mm (95% CIs, 1.01–1.21), P = 0.034]. Similar results were obtained in Cox models with SUA levels partitioned according to baseline quartiles and in which AF was defined by physician reports or by both Minnesota coding and physician reports.
Conclusions:
In-treatment SUA is a strong predictor for new-onset AF in hypertensive patients, independent of effects of antihypertensive treatment, age, sex, and ECG-LVH. Further research is needed to clarify how uric acid may provoke AF (ClinicalTrials.gov identifier: NCT00338260).
DOI: https://doi.org/10.37349/emed.2022.00079
This article belongs to the special issue Angiotensins—A Century of Progress

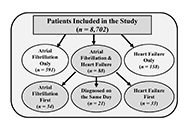
Aim:
The present study investigated the appearance and severity of atrial fibrillation (AF) and heart failure (HF) in 8,702 hypertensive patients with left ventricular hypertrophy (LVH) receiving antihypertensive treatment in a prospective trial.
Methods:
Patients who had a history of AF or HF were not included, and the participants had sinus rhythm when they were randomly allocated to blinded study medication. Endpoints were adjudicated.
Results:
Incident AF occurred in 679 patients (7.8%) and HF in 246 patients (2.8%) during 4.7 ± 1.1 years mean follow-up. Incident AF was associated with a > 4-fold increased risk of developing subsequent HF [hazards ratios (HRs) = 4.7; 95% confidence intervals (CIs), 3.1–7.0; P < 0.001] in multivariable Cox analyses adjusting for age, sex, race, randomized treatment, standard cardiovascular risk factors and incident myocardial infarction. The development of HF as a time-dependent variable was associated with a multivariable-adjusted 3-fold increase of the primary study endpoint (HRs = 3.11; 95% CIs, 1.52–6.39; P < 0.001) which was a composite of myocardial infarction, stroke or cardiovascular death. Incident HF was associated with a > 3-fold increased risk of developing subsequent AF (HRs = 3.3; 95% CIs, 2.3–4.9; P < 0.001). This development of AF was associated with a > 2-fold increase of the composite primary study endpoint in multivariable Cox analysis (HRs = 2.26; 95% CIs, 1.09–4.67; P = 0.028).
Conclusions:
Incident atrial fibrillation and heart failure are associated with increased risk of the other in treated hypertensive patients with left ventricular hypertrophy. Such high-risk hypertensive patients who subsequently develop both atrial fibrillation and heart failure have particular high risk of composite myocardial infarction, stroke or cardiovascular death (ClinicalTrials.gov identifier: NCT00338260).
Aim:
The present study investigated the appearance and severity of atrial fibrillation (AF) and heart failure (HF) in 8,702 hypertensive patients with left ventricular hypertrophy (LVH) receiving antihypertensive treatment in a prospective trial.
Methods:
Patients who had a history of AF or HF were not included, and the participants had sinus rhythm when they were randomly allocated to blinded study medication. Endpoints were adjudicated.
Results:
Incident AF occurred in 679 patients (7.8%) and HF in 246 patients (2.8%) during 4.7 ± 1.1 years mean follow-up. Incident AF was associated with a > 4-fold increased risk of developing subsequent HF [hazards ratios (HRs) = 4.7; 95% confidence intervals (CIs), 3.1–7.0; P < 0.001] in multivariable Cox analyses adjusting for age, sex, race, randomized treatment, standard cardiovascular risk factors and incident myocardial infarction. The development of HF as a time-dependent variable was associated with a multivariable-adjusted 3-fold increase of the primary study endpoint (HRs = 3.11; 95% CIs, 1.52–6.39; P < 0.001) which was a composite of myocardial infarction, stroke or cardiovascular death. Incident HF was associated with a > 3-fold increased risk of developing subsequent AF (HRs = 3.3; 95% CIs, 2.3–4.9; P < 0.001). This development of AF was associated with a > 2-fold increase of the composite primary study endpoint in multivariable Cox analysis (HRs = 2.26; 95% CIs, 1.09–4.67; P = 0.028).
Conclusions:
Incident atrial fibrillation and heart failure are associated with increased risk of the other in treated hypertensive patients with left ventricular hypertrophy. Such high-risk hypertensive patients who subsequently develop both atrial fibrillation and heart failure have particular high risk of composite myocardial infarction, stroke or cardiovascular death (ClinicalTrials.gov identifier: NCT00338260).
DOI: https://doi.org/10.37349/emed.2022.00080
This article belongs to the special issue Angiotensins—A Century of Progress

Aim:
Whether incident left bundle branch block (LBBB) is associated with increased cardiovascular (CV) morbidity and mortality in treated hypertensive patients with left ventricular hypertrophy (LVH) is unknown. Thus, the present study aimed to examine CV outcomes of incident LBBB in treated hypertensive patients with LVH.
Methods:
In the Losartan Intervention For Endpoint reduction in hypertension (LIFE) study, 9,193 hypertensive patients with LVH on screening electrocardiogram (ECG) were randomized to losartan or atenolol based treatment. Participants (n = 8,567) did not have LBBB (Minnesota code 7.1) on baseline ECG. Cox regression models controlling for significant covariates assessed independent associations of incident LBBB with CV events and all-cause mortality during 4.8 years mean follow-up.
Results:
Annual follow-up ECGs identified 295 patients (3.4%) with incident LBBB associated with male gender (P < 0.05), older age, higher Cornell voltage (both P < 0.005) and history of diabetes, isolated systolic hypertension and prevalent CV disease. When adjusted for the history of previous CV disease, diabetes, isolated systolic hypertension, the Framingham risk score, ECG-LVH and randomized study treatment, Cox regression models showed that incident LBBB predicted higher risk of the composite endpoint CV death, myocardial infarction and stroke [hazard ratio (HR) 1.9, 95% confidence intervals (CIs) 1.3–2.9, P < 0.001], CV death (HR 3.0, 95% CIs 1.84–5.0, P < 0.001), heart failure (HR 3.6, 95% CIs 1.9–6.6, P < 0.001) and all-cause mortality (HR 3.0, 95% CIs 2.0–4.3, P < 0.001).
Conclusions:
These data suggest that among hypertensive patients with ECG-LVH receiving aggressive antihypertensive therapy, incident LBBB independently predicts increased risk of subsequent CV events including congestive heart failure and CV and all-cause mortality (ClinicalTrials.gov identifier: NCT00338260).
Aim:
Whether incident left bundle branch block (LBBB) is associated with increased cardiovascular (CV) morbidity and mortality in treated hypertensive patients with left ventricular hypertrophy (LVH) is unknown. Thus, the present study aimed to examine CV outcomes of incident LBBB in treated hypertensive patients with LVH.
Methods:
In the Losartan Intervention For Endpoint reduction in hypertension (LIFE) study, 9,193 hypertensive patients with LVH on screening electrocardiogram (ECG) were randomized to losartan or atenolol based treatment. Participants (n = 8,567) did not have LBBB (Minnesota code 7.1) on baseline ECG. Cox regression models controlling for significant covariates assessed independent associations of incident LBBB with CV events and all-cause mortality during 4.8 years mean follow-up.
Results:
Annual follow-up ECGs identified 295 patients (3.4%) with incident LBBB associated with male gender (P < 0.05), older age, higher Cornell voltage (both P < 0.005) and history of diabetes, isolated systolic hypertension and prevalent CV disease. When adjusted for the history of previous CV disease, diabetes, isolated systolic hypertension, the Framingham risk score, ECG-LVH and randomized study treatment, Cox regression models showed that incident LBBB predicted higher risk of the composite endpoint CV death, myocardial infarction and stroke [hazard ratio (HR) 1.9, 95% confidence intervals (CIs) 1.3–2.9, P < 0.001], CV death (HR 3.0, 95% CIs 1.84–5.0, P < 0.001), heart failure (HR 3.6, 95% CIs 1.9–6.6, P < 0.001) and all-cause mortality (HR 3.0, 95% CIs 2.0–4.3, P < 0.001).
Conclusions:
These data suggest that among hypertensive patients with ECG-LVH receiving aggressive antihypertensive therapy, incident LBBB independently predicts increased risk of subsequent CV events including congestive heart failure and CV and all-cause mortality (ClinicalTrials.gov identifier: NCT00338260).
DOI: https://doi.org/10.37349/emed.2022.00081
This article belongs to the special issue Angiotensins—A Century of Progress

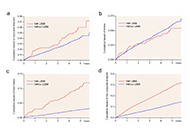
Aim:
While it is commonly thought that left ventricular (LV) systolic function may insidiously deteriorate in hypertensive patients, few prospective data are available to support this notion.
Methods:
We evaluated 680 hypertensive patients (66 ± 7 years; 45% women) with electrocardiographic (ECG)-LV hypertrophy (ECG-LVH) enrolled in the Losartan Intervention For Endpoint reduction in hypertension (LIFE) echo-sub-study free of prevalent cardiovascular disease and with baseline ejection fraction (EF) ≥ 55%. Echocardiographic examinations were performed annually for 5 years during anti-hypertensive treatment. Development of reduced systolic function was defined as incident EF < 50%.
Results:
During a mean follow-up of 4.8 ± 1 years, 37 patients developed reduced EF without an inter-current myocardial infarction (5.4%). In analysis of covariance, patients who developed reduced EF were more often men, had greater baseline LV diameter and LV mass, lower mean EF (all P < 0.05), and similar diastolic function indices. At the last available examination before EF reduction, independently of covariates, patients with reduced EF showed a significant increase in left atrium (LA) size, LV diameter, end-systolic stress and mitral E/A ratio, as compared to those who did not develop reduced EF (all P < 0.05). In time-varying Cox regression analysis, also controlling for baseline EF, predictors of developing reduced EF were higher in-treatment LV diameter [hazard ratio (HR) = 5.19 per cm; 95% confidence interval (CI): 2.58–10.41] and higher in-treatment mitral E/A ratio (HR = 2.37 per unit; 95% CI: 1.58–3.56; both P < 0.0001).
Conclusions:
In treated hypertensive patients with ECG-LVH at baseline, incident reduced EF is associated with the development of dilated LV chamber and signs of increased LV filling pressure (ClinicalTrials.gov identifier: NCT00338260).
Aim:
While it is commonly thought that left ventricular (LV) systolic function may insidiously deteriorate in hypertensive patients, few prospective data are available to support this notion.
Methods:
We evaluated 680 hypertensive patients (66 ± 7 years; 45% women) with electrocardiographic (ECG)-LV hypertrophy (ECG-LVH) enrolled in the Losartan Intervention For Endpoint reduction in hypertension (LIFE) echo-sub-study free of prevalent cardiovascular disease and with baseline ejection fraction (EF) ≥ 55%. Echocardiographic examinations were performed annually for 5 years during anti-hypertensive treatment. Development of reduced systolic function was defined as incident EF < 50%.
Results:
During a mean follow-up of 4.8 ± 1 years, 37 patients developed reduced EF without an inter-current myocardial infarction (5.4%). In analysis of covariance, patients who developed reduced EF were more often men, had greater baseline LV diameter and LV mass, lower mean EF (all P < 0.05), and similar diastolic function indices. At the last available examination before EF reduction, independently of covariates, patients with reduced EF showed a significant increase in left atrium (LA) size, LV diameter, end-systolic stress and mitral E/A ratio, as compared to those who did not develop reduced EF (all P < 0.05). In time-varying Cox regression analysis, also controlling for baseline EF, predictors of developing reduced EF were higher in-treatment LV diameter [hazard ratio (HR) = 5.19 per cm; 95% confidence interval (CI): 2.58–10.41] and higher in-treatment mitral E/A ratio (HR = 2.37 per unit; 95% CI: 1.58–3.56; both P < 0.0001).
Conclusions:
In treated hypertensive patients with ECG-LVH at baseline, incident reduced EF is associated with the development of dilated LV chamber and signs of increased LV filling pressure (ClinicalTrials.gov identifier: NCT00338260).
DOI: https://doi.org/10.37349/emed.2022.00082
This article belongs to the special issue Angiotensins—A Century of Progress

